1. FOREWORD AND CNIC MISSION
Valentín Fuster
General DirectorVicente Andrés
Basic Research DirectorBorja Ibáñez
Clinical Research DirectorThe CNIC’s mission is to discover the causes of cardiovascular disease (CVD), translate basic research discoveries into clinical practice, promote health in society, and foster training and mentoring of up-and-coming scientists and physicians. In 2019, 415 researchers and technicians carried out their scientific activity at the CNIC. These scientists are members of 31 Research Groups and 11 Technical Units distributed across three multidisciplinary Research Areas: Vascular Physiopathology, Myocardial Physiopathology, and Cell Developmental Biology. These areas constitute and connect the CNIC’s two research departments: Basic Research and Clinical Research.
Over its relatively short existence, the CNIC has built an unrivalled infrastructure and a powerful, cross-disciplinary research base that embraces many disciplines and includes population and patient studies. A key factor in this achievement is the commitment of the Spanish government to building a flagship research institute to tackle the CVD epidemic. Most importantly, the CNIC is supported through an innovative public–private initiative financed by the Carlos III Institute of Health and the Pro CNIC Foundation. The CNIC also benefits from the external support and advice of its recently renewed Scientific Advisory Board, composed of leading international experts who provide guidance on strategy and recruitment and regularly evaluate the performance of the Center and its group leaders. In 2019, Rodrigo Fernández and Maria A. Moro joined the Center as Assistant Health Scientist and Full Professor, respectively.
At the heart of the CNIC’s mission is a shift from the traditional emphasis on treating clinical events to a strategy focused on identifying CVD in its preclinical stages and promoting health. This vision fosters a cohesive and flexible strategy that embraces research infrastructure, professional training, and a clear focus on cross-disciplinary collaborations between basic and clinical researchers to ensure that acquired knowledge is translated into real health benefits.
In 2019, the CNIC was the top ranked European cardiovascular research center according to the Scimago institutions ranking, and the second ranking center worldwide, just behind the US National Heart, Lung and Blood Institute (NIH). Likewise, the CNIC was identified as one of four European Union centers of excellence in cardiovascular biomedical research by the European Research Area Network on Cardiovascular Diseases in its 2019 Strategic Research Agenda for Cardiovascular Diseases (SRA-CVD): Challenges and Opportunities for Cardiovascular Disease Research, which analyzes the current status of research in this area.
This CNIC Scientific Report offers an overview of how our young, energetic team of dedicated scientists, clinicians, and technicians is bringing our mission to reality. Reading these pages, what gives us the greatest pleasure is to see how the breadth of the CNIC’s research activity integrates the Center into society at so many levels.
As you would expect, 2019 saw breakthroughs at the frontiers of basic and clinical research. These discoveries range from studies showing delays in the appearance of Alzheimer’s disease in mice treated with an oral anticoagulant to a description of a new method to improve treatment of atrial fibrillation.
The recently launched Clinical Trials Coordination Unit has been established to enhance Spanish leadership in the field of clinical trials in the cardiovascular area. This unit is already supervising eleven clinical studies and trials. The Center’s translational studies bear testimony to the enthusiastic participation of healthy volunteers, patients, and emergency service personnel in efforts to define the causes and risk factors of CVD. This commitment of citizens and professionals outside the research community is making essential contributions to advancing the use of noninvasive imaging technology for diagnosis and research.
The CNIC’s commitment to public health promotion is also evident in training programs that start with children from an early age, teaching core health knowledge and instilling a positive emotional attitude. The Center’s public outreach links seamlessly with our strong commitment to training at all levels, from programs to encourage a scientific vocation among high school students to continuing professional training programs for scientists and physicians.
Through these endeavors, the CNIC is making a comprehensive, across-the-board investment for societal benefit that integrates biomedical research into the wider society. This is fitting, since we are all stakeholders in our health and in the health of the next generation. As we move forward, the CNIC will maintain the drive and focus established in its initial phases and ensure that the Center’s basic and clinical scientists continue to work closely together to devise innovative projects that help reduce the health care and socioeconomic burden associated with CVD.
2. Research at the Center
The CNIC is organized into two departments, one focused on Basis Research and the other on Clinical Research. Research in these fields is fully interconnected through three multidisciplinary Research Areas.
2.1 Vascular Pathophysiology
Francisco Sánchez Madrid
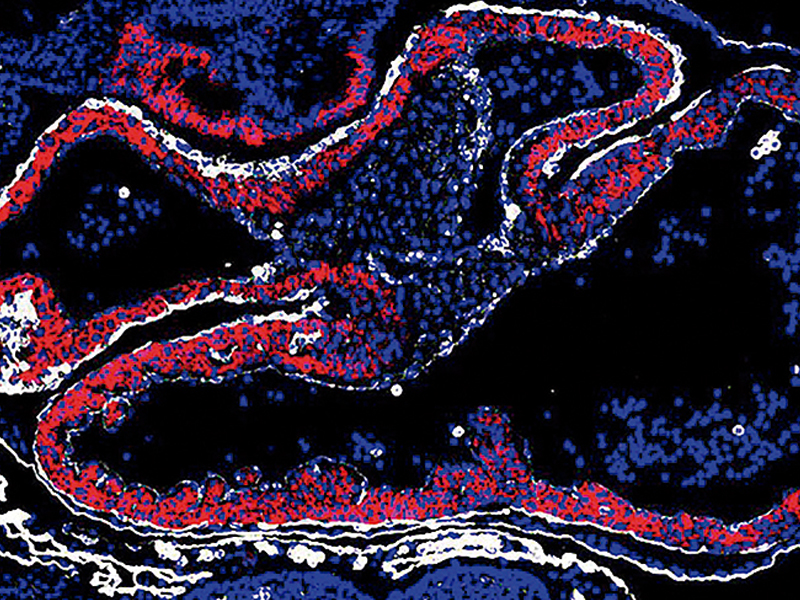
CoordinatorThe focus of the Vascular Pathophysiology Area is the biology of the vascular system in health and disease, investigated through multidisciplinary approaches that include molecular and cellular biology, animal models of disease, and translational and clinical studies. The Area hosts three technical units - Genomics, Proteomics, and Bioinformatics -, which provide state-of-the art technology to CNIC scientists while actively participating in the Area’s research projects. Our research takes advantage of high-throughput genomics, proteomics and metabolomics coupled to bioinformatic analysis, as well as state-of-the art imaging technologies, all applied to both animal models and population studies with the aim of opening up new diagnostic and prognostic avenues. We conduct translational research into atherosclerosis, the main underlying cause of heart attack and stroke. We are interested in dissecting the regulatory pathways involved in vessel wall and cardiac remodeling and the contribution of aging to these events, including both physiological and premature aging. We also investigate the role of smooth muscle and endothelial cells and the interplay between the hematopoietic and cardiovascular systems during atherosclerosis development, as well as the signaling pathways that regulate cardiovascular development and disease and age-associated alterations of muscle cells that could account for the decline in tissue regeneration with age. Another important area of interest is the immune and inflammatory component of cardiovascular disease, including the role in atherosclerosis of somatic mutation-driven clonal hematopoiesis and the antibody immune response, the mechanisms of intercellular communication between immune cells, and the role of T cell and immunomodulatory molecules in the development of myocarditis.
TECHNICAL UNITS
RESEARCH GROUPS
2.2 Cell and deVeloPMental Biology
Miguel Ángel del Pozo
Coordinator
The Cell and Developmental Biology (CDB) Area comprises eight research groups and three technical units devoted to basic studies and their translational projection in vascular development, homeostasis, and disease. Some groups seek to understand how the spatiotemporal regulation of genome architecture and expression determines cell decisions in the early embryo and heart development, thus contributing to the advancement of cardiovascular (CV) regenerative medicine. Other groups investigate cell and tissue mechanisms that determine CV function, such as angiogenesis, inflammation, and repair, and explore the principles controlling the mechanical function and adaptability of the CV system. This research line deploys multidisciplinary programs integrating cell and systems biology, biophysics, and single-molecule techniques. Efforts are specifically devoted to building bridges between basic research and cardiovascular medicine, with a focus on cardiomyopathies, atherosclerosis, and cerebrovascular disease.
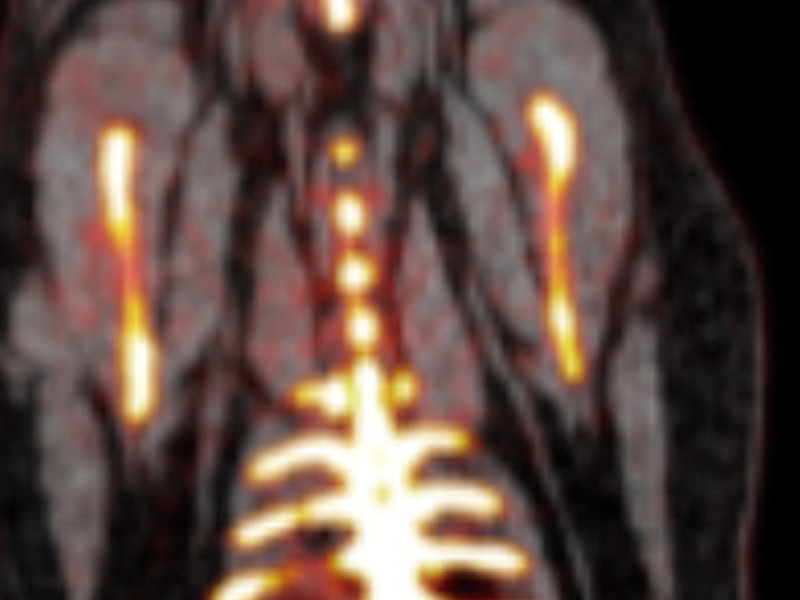
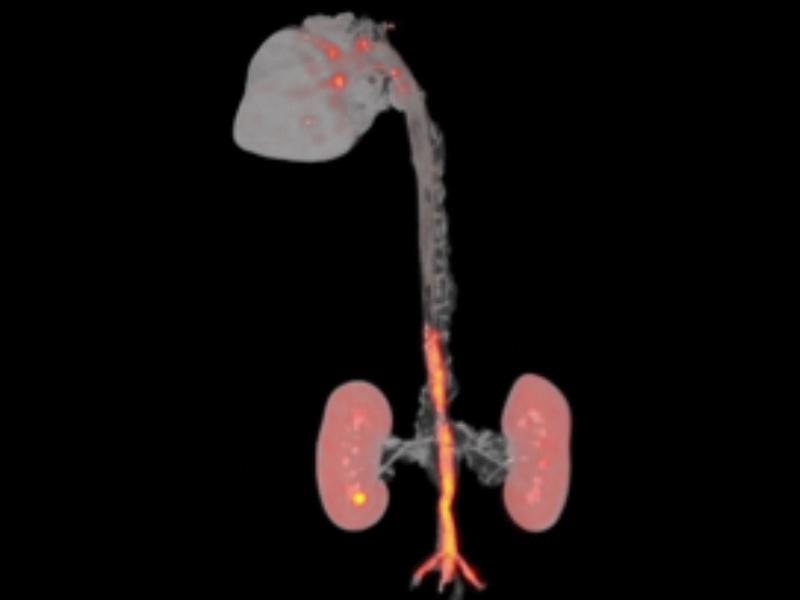
The CDB's three core technical units provide support on state-of-the-art visualization techniques and develop solutions covering different scales and biological processes. The Microscopy Unit offers advanced confocal, multiphoton, and super-resolution imaging technologies, together with approaches for quantitative biology. The Cellomics Unit provides cytometry and separation services (including state-of-the-art spectral cytometry), as well as a high-content functional genomics screening platform. Both units provide support for tailored image analysis and data processing. The Advanced Imaging Unit offers a portfolio of cutting- edge preclinical imaging services for small animals (ultrasound, magnetic resonance, PET/CT, optical imaging, and radiochemistry). The unit provides support to the Center’s research groups in the assessment of various animal models and performs its own technical research on advanced molecular imaging techniques. The Advanced Imaging Unit was awarded ISO 9001:2015 quality accreditation in June 2018.
TECHNICAL UNITS
RESEARCH GROUPS
2.3 Myocardial Pathophysiology
David Sancho
Coordinator
The Myocardial Pathophysiology Area (MPA) brings together scientists from multiple disciplines.MPA groups work on several topics: the oxidative phosphorylation system, the role of nuclear receptors in lipid metabolism and inflammatory responses, metabolic syndrome and stress kinases, immunobiology, inherited cardiomyopathies, cardiac arrhythmias, cardiomyocyte electrophysiology, molecular regulation of heart failure, and translational cardiovascular imaging and therapy. Our research in these areas produced several significant advances in 2019:
(1) Development of an innovative and translational approach to identify specific atrial areas as targets for atrial fibrillation ablation.
(2) Establishment of an educational intervention for instilling healthy behaviors in children living in diverse and socioeconomically disadvantaged communities.
(3) New findings on how nuclear receptors control tissue resident macrophage identity by regulating chromatin accessibility and the transcription of canonical macrophage genes, and how macrophages communicate with endothelial cells to promote cardiac repair and remodeling.
(4) The use of adeno-associated virus-mediated gene transfer in mice, proteomics, and CRISPR/Cas9 gene editing in human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) to investigate the emerging role of NaV1.5-Kir2.1 macromolecular ion channel complexes in the mechanisms of sudden cardiac death in inheritable diseases.
(5) Studies of the role of RNA-binding proteins in the adult heart and the molecular mechanisms underlying the development of arrhythmogenic cardiomyopathy (AC) type 5.
(6) Work on the role of the OxPhos system in health and disease, highlighting the role of mitochondrial ROS in OxPhos system adaptation to cellular metabolic requirements.
(7) New advances in deciphering the mechanisms involved in Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT) and Long QT Syndrome type 8.
(8) Analysis of how host-microbiota interactions regulate the immune gut barrier, showing that microbiota dysbiosis and translocation might regulate low-grade systemic inflammation underlying metabolic syndrome and cardiovascular disease.
(9) Studies of p38gamma showed that this kinase is involved in the progression from steatosis to liver cancer and in the control of the proliferation of hepatocytes after stress. In addition, stress in adipose tissue is an important predisposing factor for liver cancer development. Interestingly, testosterone increases adipose tissue stress, reflecting the higher incidence of liver cancer in men than women.
TECHNICAL UNITS
RESEARCH GROUPS
The MPA’s core technical units give support to all CNIC scientists: 1) The Transgenesis Unit provides services in mouse strain rederivation, production of genetically modified mice, and cryopreservation of mouse strains; 2) the Pluripotent Cell Technology Unit has generated an isogenic hiPSC line and optimized differentiation protocols to model CVD using hiPSC-derived cardiomyocytes; 3) the Clinical Trials Coordination Unit continues with its mission to boost Spanish leadership in clinical trials in the cardiovascular area; 4) the Viral Vectors Unit provides researchers with access to state-of-the-art viral vector technology for use in preclinical studies and basic research applications; 5) the Comparative Medicine Unit supports in vivo work in the animal facility.
2.4 Clinical Studies
Progression of Early Subclinical AtherosclerosiS
(PESA - CNIC - Santander)

Principal investigator:
Valentín Fuster
The PESA-CNIC-Santander study is a long-term endeavor carried out by the CNIC in collaboration with Santander Bank. This study aims to identify the presence of atherosclerosis long before symptoms appear and to define the cues leading to its development and progression. The study, led by CNIC General Director Valentin Fuster, launched in 2010 and enrolled 4184 asymptomatic individuals between the ages of 40 and 55. Participants undergo serial (every 3 years) imaging and analytical tests, including 3D vascular ultrasound of the carotid arteries, aorta, and iliofemoral arteries to detect atherosclerotic plaques, coronary artery calcium quantification by computed tomography, and biosampling for omics analysis. A subset of 946 participants showing signs of disease are undergoing vascular 18FDG PET/MR and cardiac MR. Several CNIC clinical and basic research groups participate in PESA, which is the Center’s flagship study. The PESA-CNIC-Santander study is already making seminal contributions to our understanding of the origin and progression of atherosclerosis.
In 2019, the third follow-up assessments of PESA participants were completed, and now in 2020 the new PESA-Health Initiative is expanding the scientific approaches performed in the continuing follow-up of the PESA cohort.
Secondary Prevention of Cardiovascular Disease in the Elderly Population
(SECURE)

Principal investigator:
Valentín Fuster
Co- Principal investigator:
José Mª Castellano
Adherence to treatment after an acute myocardial infarction (MI) is essential for efficient secondary prevention. Despite this, many post-MI patients abandon prescribed medication. To address this issue, CNIC researchers and FERRER laboratories developed a “polypill” including three key drugs prescribed to post-MI patients (aspirin, an ACE-inhibitor, and a statin). Having demonstrated that prescription of the CNIC Polypill significantly increases treatment adherence among post-MI patients (J Am Coll Cardiol. 2014; 64:2071-82), CNIC researchers are now leading a multinational randomized clinical trial supported by the H2020 programme. The ongoing SECURE trial (trial identifier NCT02596126) has enrolled 2500 patients soon after an MI and randomized them to standard treatment or a CNIC Polypill-based strategy. Patients will be followed up for a minimum of 2 years, and the incidence of major cardiovascular events will be assessed. Trial enrollment was completed by the end of 2019.
This trial has been extended until December 2021, and is now in the follow-up phase.
The TANSNIP-PESA randomized control trial
(TANSNIP)
a 30-month worksite-based lifestyle program to promote cardiovascular health in middle aged bank employees

Principal investigator:
Valentín Fuster
Existing tools for characterizing atherosclerosis and determining the risk of its complications are inadequate. These deficiencies limit effective management across the spectrum of this disease, and therefore opportunities are lost for early, cost-effective interventions in subclinical disease, while high-risk populations with manifest disease are administered treatments almost indiscriminately. This leads to high ‘numbers needed-to-treat’ (NNT), unnecessary patient risk, wasted resources, and unsustainable costs for health care purchasers. In a relatively low-risk population (the PESA-CNIC cohort), we will study whether a personalized worksite-based lifestyle intervention, driven by imaging data (2D and 3D-ultrasound of carotid and ilio-femoral arteries plus coronary artery calcification) results in changes in behavior, improved control of risk factors, and reduced progression of subclinical atherosclerosis plaque burden (SAPB). TANSNIP is a randomized control trial (RCT) including middle–aged bank employees from the PESA cohort stratified by SAPB (high SABP n=260; low SABP n= 590). Within each stratum, participants are randomized 1:1 to join a lifestyle program or receive standard care. The program consists of three elements: (1) 12 personalized lifestyle counseling sessions using motivational interviewing over a 30-month period, (2) a wrist-worn physical activity tracker, and (3) a sit-stand workstation. The primary outcome measure is a composite score of blood pressure (BP), physical activity, sedentary time, body weight, diet, and smoking (the adapted FUSTER-BEWAT score), measured at baseline and at 1-, 2-, and 3-year follow-up. Secondary outcomes are individual changes in lifestyle behaviors and specific changes in anthropometric measures, blood biomarkers, self-rated health, work-related outcomes (including work productivity and absenteeism), health care consumption, program process measures, and cost measures at different measurement points.
The expectation is that individual awareness of CVD risk stratification in the intervention group will lead to a reduction in the prevalence of CV risk factors related to lifestyle and an increase in physical activity compared with the control group. A second rationale is that the level of compliance with the comprehensive 3-year worksite-based lifestyle intervention will be higher among participants with a high imaging defined CV risk.
The trial has been finished and primary results will be reported in Q1 2021.
TREatment with Beta-blockers after myOcardial infarction (MI) withOut reduced ejection fracTion
(REBOOT)

Principal investigator:
Borja Ibáñez
The prescription of beta-blockers to patients after an MI is based on evidence from trials performed in the pre-reperfusion era. While there is solid evidence for their benefit in post-MI patients with reduced ejection fraction, such evidence is lacking for patients with preserved ejection fraction. Despite this, more than 80% of post-MI patients in this category are prescribed beta-blockers for the rest of their lives. REBOOT (tREatment with Beta-blockers after myOcardial infarction withOut reduced ejection fracTion) is a multinational trial that will enroll 8600 post-MI patients with a left ventricular ejection fraction above 40%. Patients are randomized to beta-blocker therapy (type and dose decided by the attending physician) or to no treatment. The primary endpoint is the composite of all-cause death, reinfarction, or heart failure admission during 3-year follow-up. This trial is coordinated by the CNIC Clinical Trials Coordination Unit and is run in close collaboration with the Mario Negri Institute of Research in Milan. 105 hospitals across Spain and Italy participate in this large- scale project that will have a major impact on clinical practice.
The first patients were enrolled in October 2018, and so far 3700 patients have been recruited. It is anticipated that enrollment will be completed before the end of 2022.
AtheroBrain. The Heart to Head STUDY
(H2H)

Principal investigator:
Valentín Fuster
Co- Principal investigator:
Héctor Bueno
There is increasing awareness of the association between atherosclerosis and cognitive function, but the mechanisms linking these processes are not fully understood. The Heart-to-Head (H2H) study is testing the hypothesis that extensive subclinical atherosclerosis is associated with subtle cognitive decline and beta-amyloid deposition in the brain. This transatlantic collaboration is framed within an agreement between the CNIC and Mount Sinai Hospital in New York and is led by CNIC General Director Valentin Fuster. In Spain, the H2H project is coordinated between the CNIC and 12 de Octubre Hospital. Other university hospitals (Fundación Jiménez Díaz, Clínico San Carlos, and Gregorio Marañón) participate in the project, which receives funding from the Instituto de Salud Carlos III (ISCIII) through a Proyecto Integrado de Excelencia. A total of 300 participants are undergoing extensive atherosclerosis phenotyping (multi-territory 3D vascular ultrasound and cardiac computed tomography) and thorough brain imaging (anatomical and functional magnetic resonance imaging and positron emission tomography (PET)-amyloid scan), as well as cognitive function testing.
Recruitment is complete, and follow-up visits are now being carried out.
Multimodal myocardial tissue characterization in patients with significant valvular disease
(MRVALVE)

Principal investigator:
Borja Ibáñez
The consequences of valvular heart disease on left ventricular (LV) dimensions, function, and tissue composition are important prognostic determinants. Current practice guidelines recommend surgical or percutaneous intervention in patients with significant valvular heart disease when symptoms develop or when LV remodeling or dysfunction occur. The most prevalent valvulopathies are aortic valve stenosis and mitral regurgitation. The transition from asymptomatic to symptomatic or from normal LV dimensions and function to LV dilatation/hypertrophy (LVH) and dysfunction is determined by changes in tissue composition, predominantly cardiomyocyte death, extracellular volume expansion, and fibrosis. Surgical or percutaneous intervention in severe valvular disease is currently guided by the presence of symptoms or gross anatomical and functional LV involvement (i.e. significant chamber dilatation or reduced ejection fraction). However, when these features appear, it is often too late for the intervention to restore normal cardiac function. There is therefore a need for tools able to detect myocardial involvement in valvular disease at early stages and guide treatment before overt deterioration of cardiac function.
In this project, we use a multimodality imaging approach (cardiac magnetic resonance [CMR] plus strain echocardiography) to characterize LV status in patients with significant valvular disease. We focus on 2 specific forms of valvular disease: aortic stenosis (a paradigm of LV pressure overload) and mitral regurgitation (a paradigm of LV volume overload).
CMR is the gold standard for anatomical and functional cardiac evaluation, including detection of focal fibrosis by late gadolinium enhancement (LGE) after contrast-gadolinium administration. Recent CMR advances include parametric T1/T2 mapping, absolute myocardial perfusion quantification, calculation of extracellular volume (a surrogate of diffuse fibrosis), and tagging. Using these advances, we will obtain highly accurate tissue characterization. Focal and diffuse fibrosis will be assessed by endovenous contrast using the gadolinium contrast agent, which is in routine clinical use and has an optimal safety profile. The assessment of diffuse fibrosis will additionally require the collection of blood samples to obtain the hematocrit.
Active deformation of the LV myocardium best assessed using the echocardiographic (Echo) strain technique, which can detect impairment of multidirectional strain (active deformation) despite preserved LV global function.
We correlate imaging data with functional data from the 6-minute walking test, which provides an objective assessment of functional exercise capacity. In addition, we use cardiac computed tomography (CT) as a noninvasive means to obtain information about the presence, location, and extent of calcium in the coronary arteries and valves; the CT-based calcium score is a widely used diagnostic and prognostic tool for patients with aortic stenosis.
By the end of 2019, 34 patients had been included, and we have now begun the follow up visits, with four of them now completed.
Treatment with β3 agonists in chronic pulmonary hypertension secondary to heart failure
(SPHERE- HF)

Principal investigator:
Ana García-Alvarez
Co- Principal investigator:
Borja Ibañez and Valentín Fuster
Pulmonary hypertension (PH) secondary to left heart disease (group PH) is the most common form of PH and currently lacks effective therapy. CNIC researchers have identified the β3 adrenergic receptor as a novel therapeutic target for this disease in a large animal model of PH (Basic Res Cardiol. 2016;111:49). The CNIC is currently leading a phase 2 clinical trial in which group 2 PH patients are randomized to standard therapy vs standard therapy plus a β3 selective agonist (trial identifier NCT02775539 and Nº EudraCT: 2016-002949-32). In total, 80 patients are being recruited at four Spanish hospitals and will be followed under treatment for 4 months. The study endpoints are pulmonary artery hemodynamics and the CMR profile.
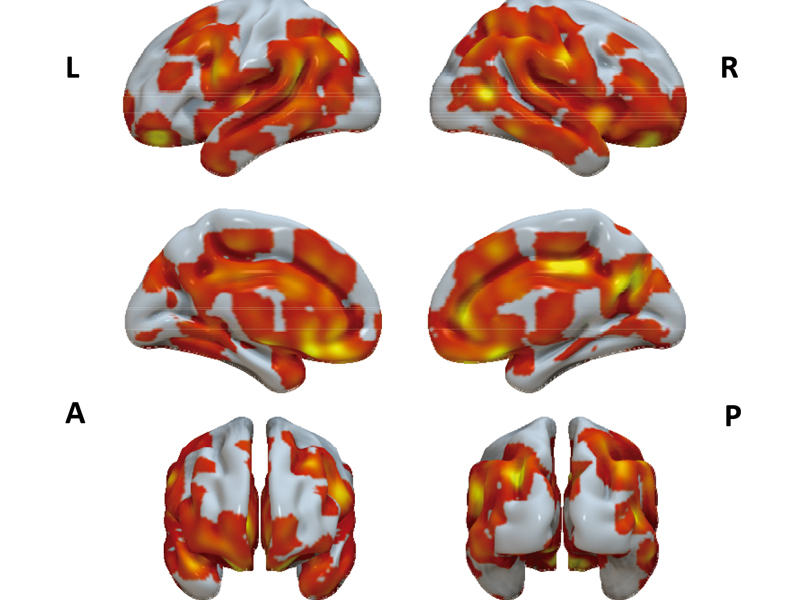
TakoTsubo Brain Project. A study to identify the Psychological and Neuro-Physiologic mechanisms of the TAKOTSUBO Syndrome
(TAKOTSUBO)

Principal investigator:
Héctor Bueno
This multicenter case-control study examines patients with takotsubo cardiomyopathy. Controls are age-, sex-, and ECG–presentation-matched AMI patients and individuals without overt CVD or acute disease.
The study includes the following substudies and techniques:
1. Psychological and neuro-psychological studies to assess personality traits, positive and negative emotions, emotional regulation, resilience, and reflection
2. EEG studies to assess the presence of neuropathohysiological responses
3. Imaging studies to determine brain areas and functional patterns related to takotsubo cardiomyopathy
4. Proteomic screening to identify molecules specifically involved in takotsubo cardiomyopathy.
T3D ULTRA-FAST COMPREHENSIVE CARDIAC MAGNETIC RESONANCE STUDY
(DASH – CMR)

Principal investigator:
Borja Ibáñez
This study includes two CMR protocols:
1) A myocardial study, including gadolinium contrast, of cardiomyopathy and myocarditis in post-infarction patients;
2) Magnetic resonance coronary angiography without gadolinium contrast in patients with a clinical indication for coronary angio-CT.
The study will include 150 patients (100 in protocol 1, 50 in protocol 2) who will undergo MRI studies at Hospital Universitario Rey Juan Carlos (3Tesla MRI) and Hospital Universitario de Salamanca (1.5Tesla MRI).
This study is funded by the ISCIII (FIS-Technological Development (FIS-DTS)). During the first year, we have validated and improved the technology in the pig model and have optimized study reconstruction so that it can be completed in real time in the MRI console.
Pulmonary vasculopathy in patients with advanced HEART failure
(HPIC)

Principal investigator colaborator:
Hospital 12 de Octubre:
Juan F Delgado
Co-Principal Investigator CNIC:
Borja Ibañez
Pulmonary hypertension due to left heart disease is a pathophysiological and hemodynamic state present in a wide range of clinical conditions that affect left heart structures. Although historically the pulmonary circulation received little attention, today it is an essential part of cardiological assessment. In patients with heart failure, the most important clinical factors are the presence of pulmonary hypertension and right ventricular function. These factors are also essential for determining prognosis and must be taken into account when making some of the most important therapeutic decisions. The pathophysiological process begins passively but later transforms into a reactive process. This reactive process includes a component that can be reversed with vasodilators and another that is fixed, in which the underlying mechanism is congestive vasculopathy (essentially medial hypertrophy and pulmonary arterial intimal fibrosis). Currently no specific therapy is available for this type of pulmonary hypertension, and treatment is the same as for heart failure itself. The drugs that have been shown to be effective in pulmonary arterial hypertension have generally shown no clear effect in clinical trials. We are clinically developing a number of groups of pharmacological compounds that will enable us to make progress in this area in the near future.
MYOCARDIAL TISSUE CHARACTERIZATION IN PATIENTS EXPOSED TO ANTHRACICLINES FOR EARLY DIAGNOSIS OF CARDIOTOXICITY
(MATRIX)

Principal investigator:
Borja Ibáñez
Cancer patients receiving anthracyclines are at risk of developing cardiac toxicity that can lead to chronic heart failure. Early diagnosis of anthracycline-induced cardiotoxicity is vital in order to implement interventons that can ameliorate heart failure progression. Current algorithms are far from ideal because cardiotoxicity is diagnosed after there is already a cardiac motion dysfuncton. Within an ongoing ERC-Consolidator project (MATRIX), we are assessing a novel, CMR-based early diagnostic test in cancer patents scheduled to undergo chemotherapy with anthracyclines. In collaboration with the Jiménez Díaz University Hospital, 100 lymphoma patents are being enrolled and will undergo serial CMR studies at the CNIC using a multiparametric approach that has been shown in a preclinical model to visualize cardiotoxicity long before the appearance of cardiac motion defects.
3. Scientific Highlights
by Publication date
Circulation
Expression of a molecule in blood cells predicts atherosclerosis risk
Earlier detection of cardiovascular disease is a step closer thanks to the findings of team of CNIC scientists led by Francisco Sánchez-Madrid and Pilar Martín. The researchers found that the expression level of the molecule CD69 in blood cells inversely predicts the appearance of subclinical atherosclerosis (developing before symptoms appear) independently of classical cardiovascular risk factors. The results, published in Circulation, show that the expression of CD69 in circulating lymphocytes correlates inversely with the presence and extent of subclinical atherosclerosis.
The origin of atherosclerosis and its progression to acute myocardial infarction and stroke involve an essential contribution from the inflammatory immune response. However, according to Francisco Sánchez-Madrid, “The relationship between lipid metabolism and the immune response is not well understood. The established hypothesis is that oxidized low-density lipoproteins (LDLox) induce the recruitment of inflammatory immune cells and their accumulation in the plaque; however, there is also evidence that cells and tissues can respond to LDLox by inhibiting proinflammatory signals.”
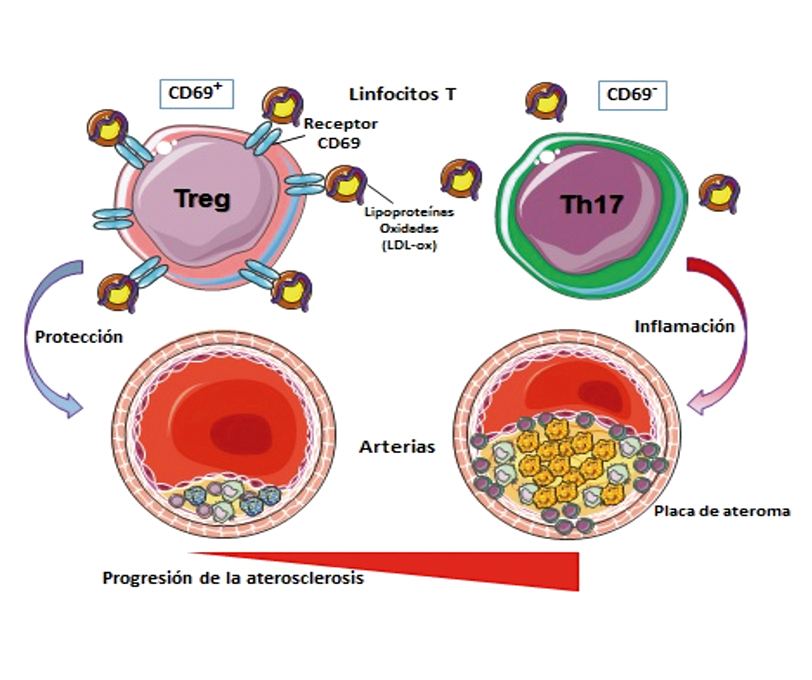
The new study identifies the molecule CD69 as a T cell receptor for oxidized lipoproteins that contributes to the control of inflammation and thus prevents the development of atherosclerosis. “Binding of LDLox to CD69 triggers the adoption of an anti-inflammatory profile by T lymphocytes that protects against the development of atherosclerosis in mice and humans,” explained Pilar Martín. The project was supported by the Spanish Cardiovascular Disease Research Network (CIBER-CV).
Tsilingiri, K., de la Fuente, H., Relano, M., Sanchez-Diaz, R., Rodriguez, C., Crespo, J., Sanchez-Cabo, F., Dopazo, A., Alonso-Lebrero, J. L., Vara, A., Vazquez, J., Casasnovas, J. M., Alfonso, F., Ibanez, B., Fuster, V., Martinez-Gonzalez, J., Martin, P., & Sanchez-Madrid, F. (2019). oxLDL Receptor in Lymphocytes Prevents Atherosclerosis and Predicts Subclinical Disease. Circulation, 139(2), 243-255. doi:10.1161/circulationaha.118.034326
JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Sleeping less than 6 hours a night increases cardiovascular risk
People who sleep fewer than 6 hours a night may be at increased risk of cardiovascular disease compared with those who sleep between 7 and 8 hours, suggest the results of the PESA CNIC-Santander Study published in the Journal of the American College of Cardiology (JACC). The study indicates that poor-quality sleep increases the risk of atherosclerosis—the build-up of plaque in the arteries throughout the body.
The researchers explained that “this new study emphasizes that we need to include sleep as one of our weapons in the fight against heart disease—a factor that is neglected in our society every day. This is the first study to show that objectively measured sleep is independently associated with atherosclerosis throughout the body, not just in the heart.”

The goal of the study, which included almost 4000 PESA CNIC-Santander participants, was to evaluate the impact of sleep duration and interrupted sleep on atherosclerosis. The study differs in several ways from previous studies on sleep and heart health. Crucially, it is larger than many earlier studies and was conducted in a healthy population; in contrast, many previous studies included people with sleep apnea or other health problems. Another important difference is that whereas other studies relied on questionnaires to determine how much sleep participants had, the new study used actimetry to obtain objective measures of sleep. As the researchers pointed out, “what people say and what they do are often different.”
Dominguez, F., Fuster, V., Fernandez-Alvira, J. M., Fernandez-Friera, L., Lopez-Melgar, B., Blanco-Rojo, R., Fernandez-Ortiz, A., Garcia-Pavia, P., Sanz, J., Mendiguren, J. M., Ibanez, B., Bueno, H., Lara-Pezzi, E., & Ordovas, J. M. (2019). Association of Sleep Duration and Quality With Subclinical Atherosclerosis. Journal of the American College of Cardiology, 73(2), 134-144. doi:10.1016/j.jacc.2018.10.060
Immunity
A new “watchdog” that controls intestinal bacteria
The immune response to our intestinal microbiota ensures that these microorganisms remain in their proper place. When the intestinal immune barrier is damaged, the gut bacteria can spread and cause inflammation throughout the body. Now, a study by scientists from the CNIC and the Universidad Complutense in Madrid reveals a new mechanism in the regulation of this immune barrier.
The study, published in Immunity, identifies a mechanism through which intestinal bacteria such as Lactobacillus strengthen the intestinal barrier to support a mutually beneficial relationship and prevent inflammation. The results have potential implications for the treatment of diseases featuring the spread of commensal bacteria outside the gut, as occurs in some metabolic disorders.
The research team propose that a new treatment strategy for these conditions could be the use of probiotics (‘beneficial’ intestinal microorganisms) or prebiotics (nutrients that promote the growth of beneficial microorganisms); however, the mechanisms underlying this strategy are so far unknown.
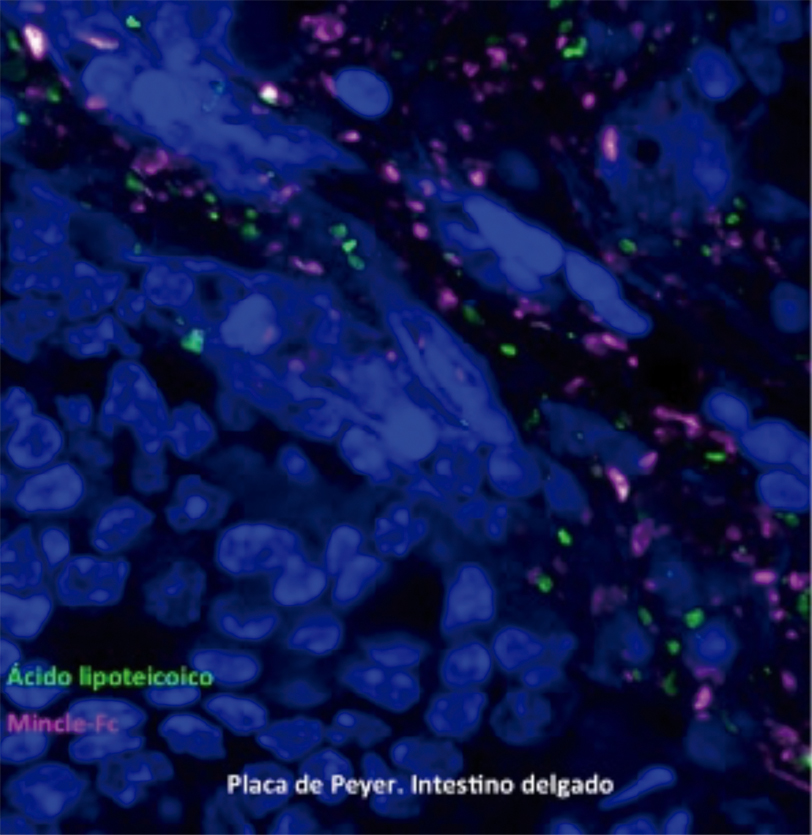
Martinez-Lopez, M., Iborra, S., Conde-Garrosa, R., Mastrangelo, A., Danne, C., Mann, E. R., Reid, D. M., Gaboriau-Routhiau, V., Chaparro, M., Lorenzo, M. P., Minnerup, L., Saz-Leal, P., Slack, E., Kemp, B., Gisbert, J. P., Dzionek, A., Robinson, M. J., Ruperez, F. J., Cerf-Bensussan, N., Brown, G. D., Bernardo, D., LeibundGut-Landmann, S., & Sancho, D. (2019). Microbiota Sensing by Mincle-Syk Axis in Dendritic Cells Regulates Interleukin-17 and -22 Production and Promotes Intestinal Barrier Integrity. Immunity, 50(2), 446-461. doi:10.1016/j.immuni.2018.12.020
Immunity
An immune “clock” that controls infections and cardiovascular disease
CNIC scientists have demonstrated the existence of an immune “clock” that coordinates the day/night cycles through the activity of a class of leucocytes called neutrophils. Neutrophils constitute the body’s main line of defense, but their action can also damage healthy cells of the cardiovascular system. This newly discovered clock determines when neutrophils are activated and when it is time to eliminate them from the circulation. First author José María Adrover explained that the researchers have identified “a series of molecules in the neutrophil nucleus and cell membrane that respond to diurnal (circadian) patterns in lightness and darkness and regulate the migration and localization of neutrophils in the body.” The study was carried out by the CNIC research group led by Andrés Hidalgo and is published in Immunity.
Due to the worldwide high prevalence of infectious and cardiovascular diseases, the study could have wide-ranging clinical implications. The team is currently working on ways to manipulate this immune clock with drugs to induce diurnal or nocturnal immunity, depending on the needs of each patient. This therapeutic approach could be valuable for people at risk of cardiovascular events and for immune-compromised patients susceptible to infection.
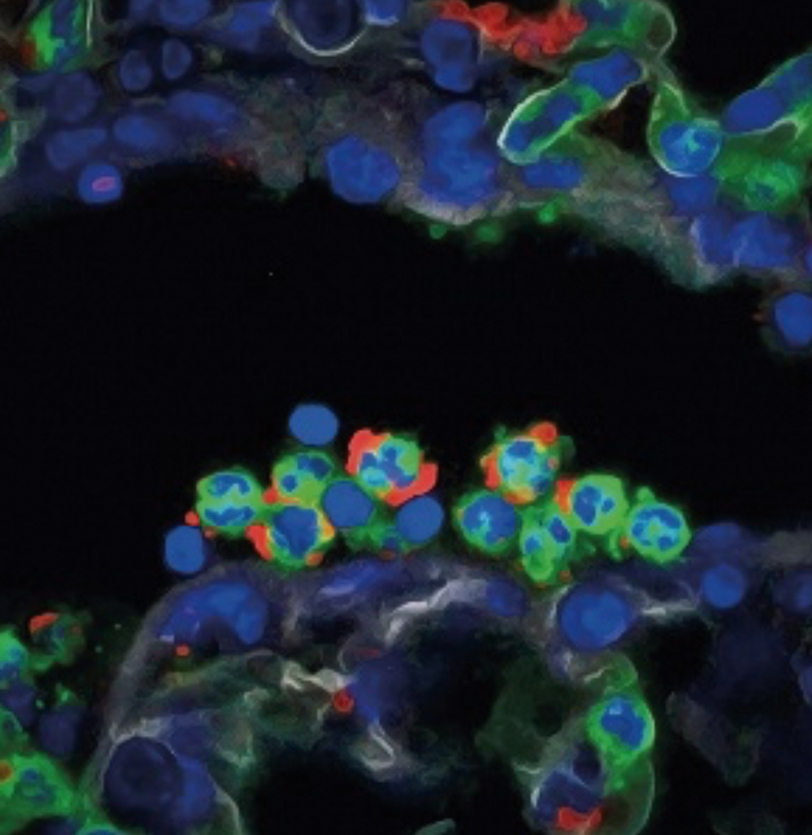
Adrover, J. M., Del Fresno, C., Crainiciuc, G., Cuartero, M. I., Casanova-Acebes, M., Weiss, L. A., Huerga-Encabo, H., Silvestre-Roig, C., Rossaint, J., Cossio, I., Lechuga-Vieco, A. V., Garcia-Prieto, J., Gomez-Parrizas, M., Quintana, J. A., Ballesteros, I., Martin-Salamanca, S., Aroca-Crevillen, A., Chong, S. Z., Evrard, M., Balabanian, K., Lopez, J., Bidzhekov, K., Bachelerie, F., Abad-Santos, F., Munoz-Calleja, C., Zarbock, A., Soehnlein, O., Weber, C., Ng, L. G., Lopez-Rodriguez, C., Sancho, D., Moro, M. A., Ibanez, B., & Hidalgo, A. (2019). A Neutrophil Timer Coordinates Immune Defense and Vascular Protection. Immunity, 50(2), 390-402. doi:10.1016/j.immuni.2019.01.002
JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
A very early marker of cardiac damage triggered by cancer treatment
Researchers at the CNIC identified a very early marker of cardiac damage in patients undergoing therapy with anthracyclines, a family of drugs commonly used to treat cancer. This finding will enable the early diagnosis of the cardiotoxicity associated with this group of widely used chemotherapy drugs.
Dr Borja Ibáñez coordinated the study, published in the Journal of the American College of Cardiology (JACC). As Dr Ibáñez explained, the results have important implications for therapy because the detection this drug-induced damage at very early stages will permit “the implementation of treatments to prevent further deterioration in heart function and a clinical management more closely adapted to the needs of each patient.” The identified marker is affected much earlier than any of the markers used in current clinical practice.
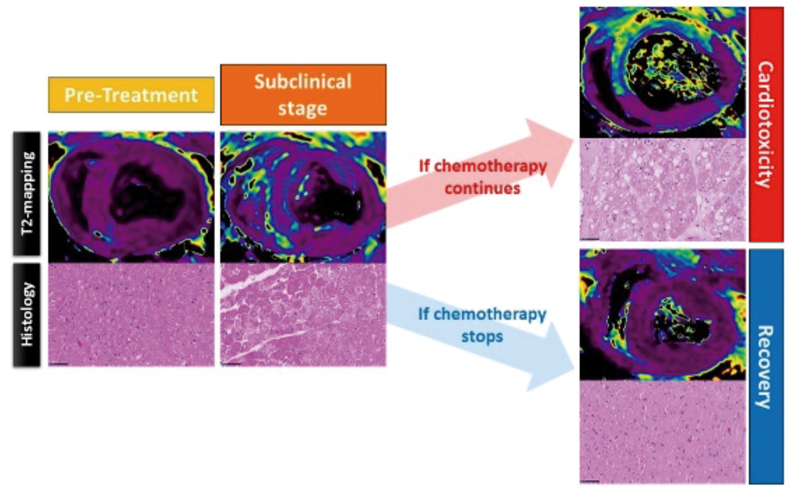
This valuable discovery was possible thanks to a new pig model of anthracycline-induced cardiotoxicity developed by the CNIC team. In the study, animals received increasing doses of the anthracycline drug doxorubicin over 10 weeks. This strategy allowed the accumulation of the drug in the heart muscle without major exposure of other organs.
The results of the JACC study may help to prevent the severe secondary effects experienced by cancer patients receiving chemotherapy. Moreover, the study may also open the way to new therapies based on mitochondrial transplantation.
Galan-Arriola, C., Lobo, M., Vilchez-Tschischke, J. P., Lopez, G. J., de Molina-Iracheta, A., Perez-Martinez, C., Aguero, J., Fernandez-Jimenez, R., Martin-Garcia, A., Oliver, E., Villena-Gutierrez, R., Pizarro, G., Sanchez, P. L., Fuster, V., Sanchez-Gonzalez, J., & Ibanez, B. (2019). Serial Magnetic Resonance Imaging to Identify Early Stages of Anthracycline-Induced Cardiotoxicity. Journal of the American College of Cardiology, 73(7), 779-791. doi:10.1016/j.jacc.2018.11.046
EMBO Molecular Medicine
New therapeutic target
for blocking early atherosclerosis in progeria
Researchers at the CNIC and the Universidad de Oviedo have discovered a new molecular mechanism involved in the premature development of atherosclerosis in mice with Hutchinson-Gilford progeria syndrome (HGPS). The results, published in EMBO Molecular Medicine, identify a potential therapeutic target for this severe genetic disease.
The study, co-directed by Vicente Andrés of the CNIC and the CIBERCV and Carlos López Otín of the Universidad de Oviedo, identifies a molecular mechanism involved in the accelerated development of atherosclerosis in progeria. In addition, the results identify a pharmacological treatment that slows the progression of atherosclerosis and extends the lifespan of progeroid mice.
The study, featured in an editorial in the journal, shows for the first time that “stress in the endoplasmic reticulum (ER stress) and the associated unfolded protein response (UPR) are involved in the death of vascular smooth muscle cells in progeroid mice,” explained first author Magda Hamczyk.
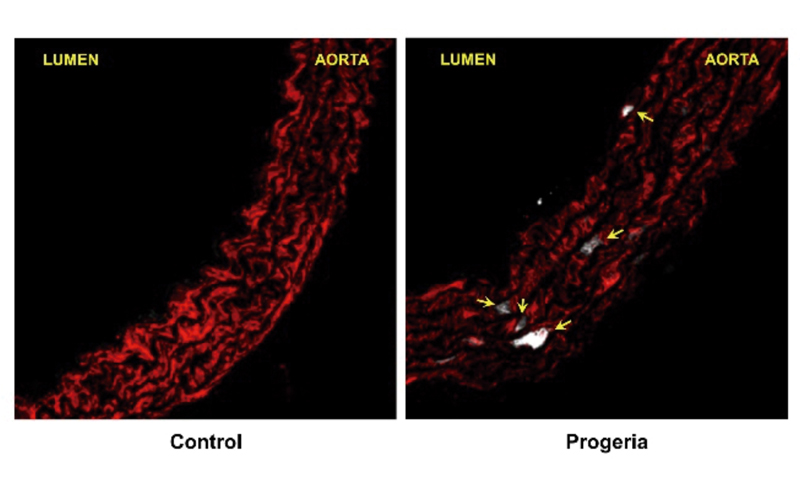
The research team used the compound tauroursodeoxycholic acid (TUDCA), which reduces the negative consequences of the activation of the ER stress and UPR pathways. Treatment of progeroid mice with TUDCA inhibits the progression of vascular disease, including vascular smooth cell loss and atherosclerosis. TUDCA also prolonged the lifespan of progeroid mice, which die from the complications of atherosclerosis.
Hamczyk, M. R., Villa-Bellosta, R., Quesada, V., Gonzalo, P., Vidak, S., Nevado, R. M., Andres-Manzano, M. J., Misteli, T., Lopez-Otin, C., & Andres, V. (2019). Progerin accelerates atherosclerosis by inducing endoplasmic reticulum stress in vascular smooth muscle cells. EMBO Molecular Medicine, 11(4), e9736. doi:10.15252/emmm.201809736
JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Advanced imaging technology predicts cardiovascular risk from inflammation detected in arteries
Using advanced PET/MRI technology, researchers at the CNIC have detected arterial inflammation in regions that have yet to develop atherosclerotic plaques. These results from the PESA-CNIC-Santander study were published in JACC. The research team used this innovative technology to analyze the inflammatory process in the arteries of a group of people who had already developed atherosclerotic plaques.
The results show that inflammation is present at early stages of atherosclerosis, above all in regions that have not developed plaques. The study also shows that this arterial atherosclerosis can be an early indication of the later appearance of plaques that underlie cardiovascular disease and events such as heart attack and stroke. The researchers are currently analyzing the role of arterial inflammation in this process; this information will help to establish early diagnosis and develop new anti-inflammatory therapies for this disease.
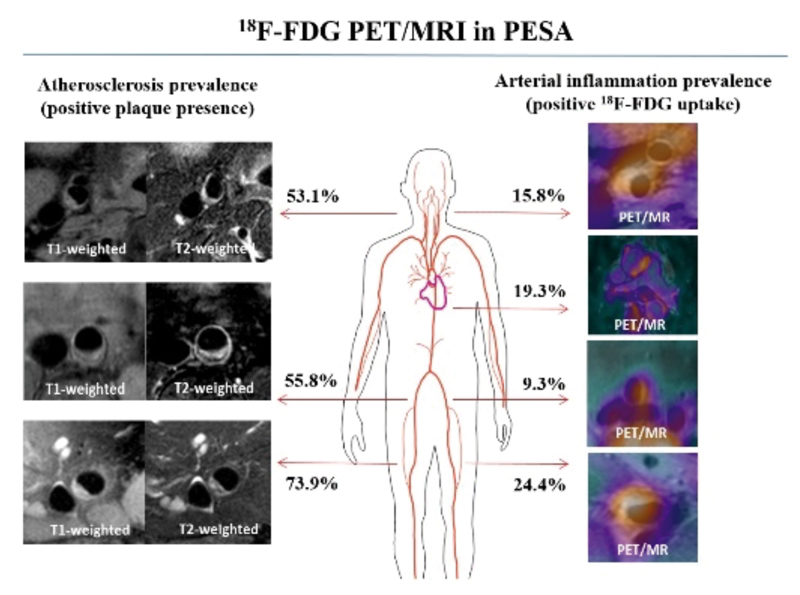
Fernandez-Friera, L., Fuster, V., Lopez-Melgar, B., Oliva, B., Sanchez-Gonzalez, J., Macias, A., Perez-Asenjo, B., Zamudio, D., Alonso-Farto, J. C., Espana, S., Mendiguren, J., Bueno, H., Garcia-Ruiz, J. M., Ibanez, B., Fernandez-Ortiz, A., & Sanz, J. (2019). Vascular Inflammation in Subclinical Atherosclerosis Detected by Hybrid PET/MRI. Journal of the American College of Cardiology, 73(12), 1371-1382. doi:10.1016/j.jacc.2018.12.075
EJournal of Experimental Medicine
Adiponectin, the hormone that protects women against liver cancer
A team at the CNIC has found an explanation for the lower rate of liver cancer in women. The answer lies in the hormone adiponectin, which is produced in higher amounts in women than in men and protects the liver against the development of the main form of liver cancer, hepatocellular carcinoma. In their quest to understand why people with obesity have a higher risk of developing liver cancer, the CNIC research group led by Guadalupe Sabio found that adiponectin is more abundant in women and slim people. The study, published in the Journal of Experimental Medicine, shows that adiponectin protects the liver against the development of hepatocellular carcinoma.
The research team showed that adiponectin, a hormone produced by adipose tissue, has an anticancer effect in the liver. In a group of healthy individuals, the team found that women produce more adiponectin than men. Describing the study, Dr. Sabio commented, “the circulating levels of adiponectin decline with the development of obesity and after puberty in men, and these are precisely the populations with higher rates of liver cancer. This observation prompted us to study the phenomenon in depth.”

The results “open routes to combating a cancer for which there is currently no specific treatment. One approach would be to use adiponectin itself, while another option is to use metformin, a drug used to treat diabetes that targets the same anticancer protein as adiponectin.”
Manieri, E., Herrera-Melle, L., Mora, A., Tomas-Loba, A., Leiva-Vega, L., Fernandez, D. I., Rodriguez, E., Moran, L., Hernandez-Cosido, L., Torres, J. L., Seoane, L. M., Cubero, F. J., Marcos, M., & Sabio, G. (2019). Adiponectin accounts for gender differences in hepatocellular carcinoma incidence. Journal of Experimental Medicine, 216(5), 1108-1119. doi:10.1084/jem.20181288
Nature
The protein p38gamma identified as a new therapeutic target in liver cancer
A research team at the CNIC led by Guadalupe Sabio has discovered that the protein p38g, one of the four types of p38 kinase, is essential for the initiation of cell division in liver cells. This indicates that “p38g could be a useful therapeutic target for liver cancer,” said Sabio, adding, “we are now developing inhibitors of this protein to test in this cancer.” The study was published in Nature. The four members of the p38 kinase family are so similar that at first they appear to have overlapping or redundant functions. Detailed analysis of their three-dimensional structures revealed that one of the four, p38g, also shares close similarities with another family of proteins called CDKs. These proteins are well-known regulators of cell division and the cell cycle and play a well-established role in the development of cancer.
The results were truly promising: “in mice lacking p38g or treated with inhibitors to block its activity, the development of hepatocellular carcinoma was slowed,” said the researchers. These results, claimed Sabio, “could be extrapolated to human patients.” Indeed, work with colleagues at Salamanca University Hospital shows that the amount of p38g increases with liver fibrosis, a process that precedes cancer and is much higher in liver cancer patients. These results suggest that in the future it may be possible to treat this type of cancer with drugs that specifically target p38g. The advantage of targeting p38g is that this enzyme appears to control the initiation of the cell cycle in response to stress, and therefore inhibiting this process would not affect tissues that are constantly proliferating, such as the intestinal lining or hair follicles.
Tomas-Loba, A., Manieri, E., Gonzalez-Teran, B., Mora, A., Leiva-Vega, L., Santamans, A. M., Romero-Becerra, R., Rodriguez, E., Pintor-Chocano, A., Feixas, F., Lopez, J. A., Caballero, B., Trakala, M., Blanco, O., Torres, J. L., Hernandez-Cosido, L., Montalvo-Romeral, V., Matesanz, N., Roche-Molina, M., Bernal, J. A., Mischo, H., Leon, M., Caballero, A., Miranda-Saavedra, D., Ruiz-Cabello, J., Nevzorova, Y. A., Cubero, F. J., Bravo, J., Vazquez, J., Malumbres, M., Marcos, M., Osuna, S., & Sabio, G. (2019). p38gamma is essential for cell cycle progression and liver tumorigenesis. Nature, 568(7735), 557-560. doi:10.1038/s41586-019-1112-8
Journal of the American College of Cardiology
Early Intervention in preschool is a unique opportunity for promoting a healthy lifestyle
Children may have a better chance of avoiding unhealthy habits linked to obesity and cardiovascular disease later in life if they are taught properly about healthy behaviors in preschool, CNIC researchers have shown in a first-of-its-kind study.
The researchers focused on children living in a socioeconomically disadvantaged community, a situation frequenly linked to higher rates of obesity, heart disease, and other health issues. Valentin Fuster created and led the trial, called the FAMILIA Project at Mount Sinai Heart. The results were published in the Journal of the American College of Cardiology.
“Results from this new study prove that early intervention is effective in preschool-age children, but we believe this can also promote healthy behaviors among their caregivers and teachers and have a far-reaching impact”, explained Dr Fuster.

This study follows other successful interventions led by Dr Fuster in Colombia and Spain, but FAMILIA is unique in being the first time the health promotion curriculum has been implemented in a multi-ethnic, underprivileged urban population. Its precedent is the Comprehensive Health Program (SI!). This intervention program is designed to promote cardiovascular health from pre-school to high school through intervention in four areas: nutrition, body and heart awareness, physical activity, and emotional management.
Fernandez-Jimenez, R., Jaslow, R., Bansilal, S., Santana, M., Diaz-Munoz, R., Latina, J., Soto, A. V., Vedanthan, R., Al-Kazaz, M., Giannarelli, C., Kovacic, J. C., Bagiella, E., Kasarskis, A., Fayad, Z. A., Hajjar, R. J., & Fuster, V. (2019). Child Health Promotion in Underserved Communities: The FAMILIA Trial. Journal of the American College of Cardiology, 73(16), 2011-2021. doi:10.1016/j.jacc.2019.01.057
Nature Communications
A newly identified mechanism can be targeted to boost angiogenesis
Scientists of the CNIC led by Rui Benedito discovered a cellular and molecular mechanism that can be exploited to induce productive and sustained angiogenesis in tissues that have become ischemic due to reduced blood supply.
Until now, tissue regeneration treatments based on vascular growth factors have not succeeded in inducing effective angiogenesis—the process through which the body generates new blood vessels. The new results, published in Nature Communications, suggest that it might be possible to manipulate the newly discovered mechanism to achieve optimal therapeutic angiogenesis.
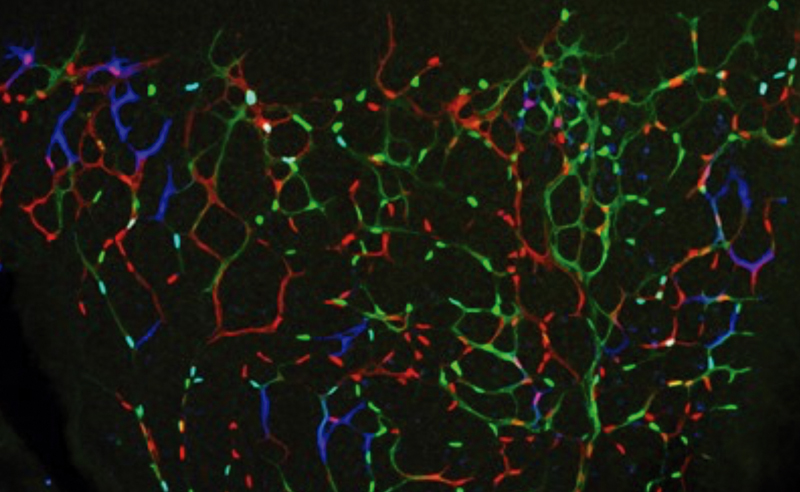
The identified mechanism could also explain the failure of several clinical trials seeking to boost angiogenesis in ischemic hearts after a myocardial infarction. Rui Benedito says that the results “significantly increase our understanding of the biology of blood vessels and will enable us to design better therapeutic strategies to induce effective angiogenesis in injured or ischemic tissues.”
Pontes-Quero, S., Fernandez-Chacon, M., Luo, W., Lunella, F. F., Casquero-Garcia, V., Garcia-Gonzalez, I., Hermoso, A., Rocha, S. F., Bansal, M., & Benedito, R. (2019). High mitogenic stimulation arrests angiogenesis. Nature Communications, 10(1), 2016. doi:10.1038/s41467-019-09875-7
Nature Communications
A new genetic tool to modify and understand gene function
Scientists of the CNIC led by Rui Benedito have developed a new genetic tool (iSuRe-Cre) that provides certainty in Cre-inducible genetic modifications, a key technique for understanding gene function.
Most analysis of gene function in biomedical research relies on the use of Cre-lox technology. Cre-Lox technology allows the regulation of gene expression at any time or in any cell type thanks to the ability of the Cre recombinase protein to recognize and recombine lox sites introduced at specific locations in the mouse genome, leading to the deletion of the genes being studied. Despite the major impact of Cre-loxP technology on biomedical research, numerous studies have demonstrated the need for caution in its use. The main problem is that the Cre activity level is often insufficient to fully recombine and eliminate expression of the target gene, generating uncertainty about whether the desired genetic modification has been achieved.
To overcome this technical hurdle, the CNIC team developed an innovative method based on a new allele called iSuRe-Cre. iSuRe-Cre is compatible with all existing Cre/CreERT2/lox alleles and guarantees high Cre activity in the cells that express the fluorescent reporter. This ultimately increases the efficiency and reliability of the analysis of Cre-dependent gene function. Moreover, the use of the new iSuRe-Cre mice permits the induction of multiple genetic deletions in the same cell. This important property allows the study of functional genetic interactions or epistasis—how the function of one or more genes depends on the function of another.
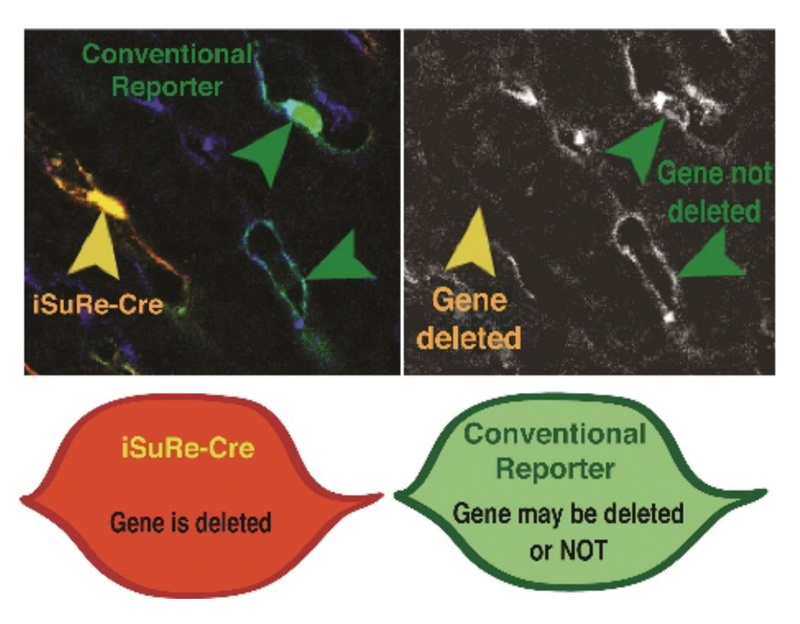
Lead investigator Rui Benedito says that the new genetic tool will be of great interest in biomedical research “because it significantly increases the ease, efficiency, and reliability of genetic modification in the mouse, the most widely used animal model in research.” The study was published in Nature Communications.
Fernandez-Chacon, M., Casquero-Garcia, V., Luo, W., Francesca Lunella, F., Ferreira Rocha, S., Del Olmo-Cabrera, S., & Benedito, R. (2019). iSuRe-Cre is a genetic tool to reliably induce and report Cre-dependent genetic modifications. Nature Communications, 10(1), 2262. doi:10.1038/s41467-019-10239-4
Circulation Research
An essential protein for correct heart
contraction and survival
A team of scientists led by Dr Enrique Lara Pezzi at the CNIC identified the RNA-binding protein SRSF3 as an essential factor for proper heart contraction and survival. In a study published in Circulation Research, the researchers found that loss of cardiac expression of SRSF3 leads to a critical reduction in the expression of genes involved in contraction. Knowledge of the mechanism of action of SRSF3 in the heart could open the way to the design of new therapeutic approaches for the treatment of heart disease. The identification of mRNA capping as a mechanism that protects against the development of systolic heart failure could open the way to the development of urgently needed therapeutic tools to combat this disease.
Ortiz-Sanchez, P., Villalba-Orero, M., Lopez-Olaneta, M. M., Larrasa-Alonso, J., Sanchez-Cabo, F., Marti-Gomez, C., Camafeita, E., Gomez-Salinero, J. M., Ramos-Hernandez, L., Nielsen, P. J., Vazquez, J., Muller-McNicoll, M., Garcia-Pavia, P., & Lara-Pezzi, E. (2019). Loss of SRSF3 in Cardiomyocytes Leads to Decapping of Contraction Related mRNAs and Severe Systolic Dysfunction. Circulation Research, 125(2), 170-183. doi:10.1161/CIRCRESAHA.118.314515
Journal of the American College of Cardiology
CNIC coordinates an international consensus document on the use of magnetic resonance imaging after a heart attack
The CNIC coordinated the first international consensus document providing guidelines on the conduct of magnetic resonance imaging studies after a myocardial infarction in clinical trials or experimental models. The document concludes that the main outcome parameter in studies assessing new treatments should be absolute infarct size—the percentage of the left ventricle that is irreversibly damaged. The recommended timing for magnetic resonance imaging is between 3 and 7 days after the infarction.
The document, coordinated by Dr Borja Ibáñez, addresses the need within the cardiovascular community for guidance on the best protocols, the best techniques, and the most appropriate situations for conducting a magnetic resonance imaging study after a heart attack. The document was published in the Journal of the American College of Cardiology (JACC).

The document’s contents were defined during an international meeting held at the CNIC with support from Philips. The meeting brought together a multidisciplinary group of 16 experts in the field from the USA, Canada, the UK, France, Germany, Sweden, the Netherlands, Greece, Switzerland, Singapore, and Spain, including Dr David García-Dorado of the CIBERCV.
Ibanez, B., Aletras, A. H., Arai, A. E., Arheden, H., Bax, J., Berry, C., Bucciarelli-Ducci, C., Croisille, P., Dall’Armellina, E., Dharmakumar, R., Eitel, I., Fernandez-Jimenez, R., Friedrich, M. G., Garcia-Dorado, D., Hausenloy, D. J., Kim, R. J., Kozerke, S., Kramer, C. M., Salerno, M., Sanchez-Gonzalez, J., Sanz, J., & Fuster, V. (2019). Cardiac MRI Endpoints in Myocardial Infarction Experimental and Clinical Trials: JACC Scientific Expert Panel. Journal of the American College of Cardiology, 74(2), 238-256. doi:10.1016/j.jacc.2019.05.024
Circulation
Possible treatment breakthrough for the rare disease arrhythmogenic right ventricular cardiomyopathy type 5
Scientists at the CNIC and Puerta de Hierro Majadahonda Hospital have discovered a possible treatment for arrhythmogenic right ventricular cardiomyopathy type 5 (ARVC5), a fatal genetic disease for which there is unfortunately no cure. The research team, whose findings were published in Circulation, showed that strategies to inhibit the protein kinase GSK3 in transgenic mice with ARVC5 reduce fibrosis and improve heart function.
The research team tested several candidate therapeutic approaches in the mouse ARVC5 model. While treatments directly targeting fibrosis were ineffective, positive results were obtained with two strategies for inhibiting GSK3, one based on pharmacological inhibition and the other on overexpression of the calcineurin subunit CnAβ1.
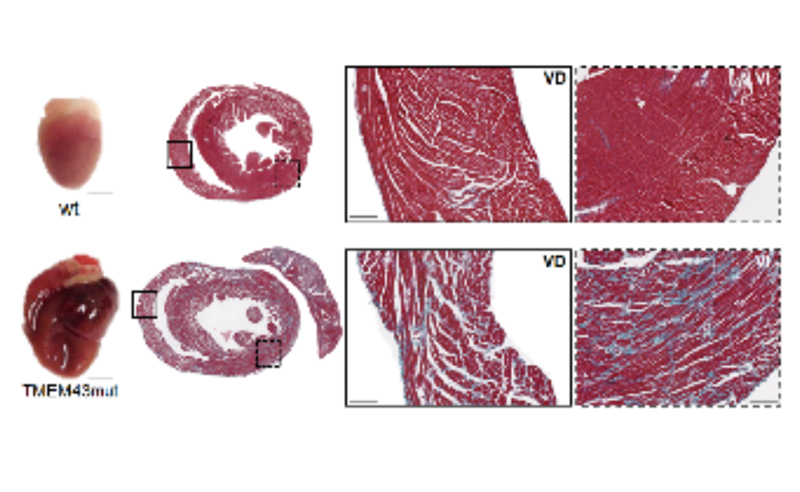
Nevertheless, the scientists warn that the transgenic mouse model does not reproduce all disease characteristics. For example, male and female mice are equally affected, whereas the human disease is much more aggressive in men than in women. Having identified a possible route for effective treatment for the disease in mice, the research team is now working to translate the results to patients. Using the mouse model, the scientists are testing drugs used to treat human heart failure to see if they are effective against ARVC5. The team is also investigating gene therapy strategies that could improve heart function or even cure the disease.
Padron-Barthe, L., Villalba-Orero, M., Gomez-Salinero, J. M., Dominguez, F., Roman, M., Larrasa-Alonso, J., Ortiz-Sanchez, P., Martinez, F., Lopez-Olaneta, M., Bonzon-Kulichenko, E., Vazquez, J., Marti-Gomez, C., Santiago, D. J., Prados, B., Giovinazzo, G., Gomez-Gaviro, M. V., Priori, S., Garcia-Pavia, P., & Lara-Pezzi, E. (2019). Severe Cardiac Dysfunction and Death Caused by Arrhythmogenic Right Ventricular Cardiomyopathy Type 5 Are Improved by Inhibition of Glycogen Synthase Kinase-3beta. Circulation, 140(14), 1188-1204. doi:10.1161/CIRCULATIONAHA.119.040366
Circulation Research
A new method to improve treatment
of atrial fibrillation
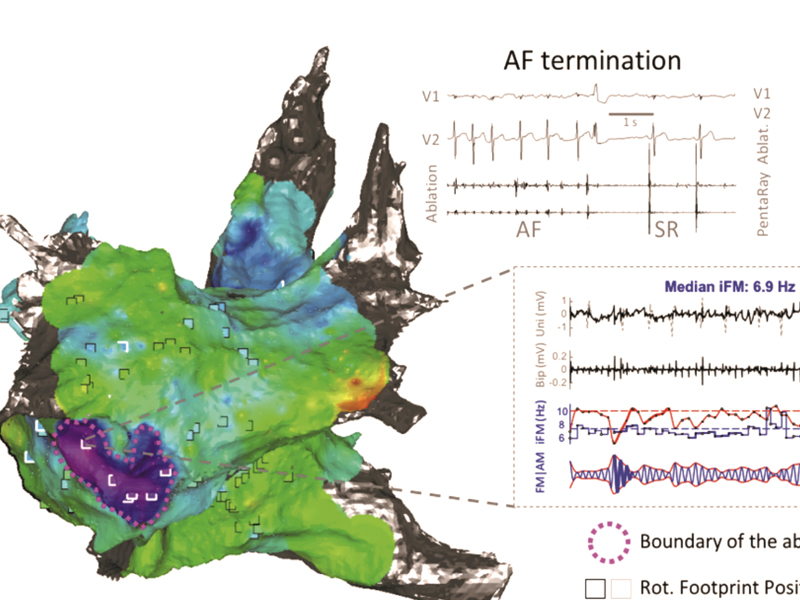
Researchers at the CNIC, the Hospital Clínico San Carlos in Madrid, and the Spanish cardiovascular research network (CiberCV) discovered a new method to optimize ablation of atrial fibrillation (AFib), one of the most common forms of irregular heartbeat (arrhythmia). The study was featured on the cover of the journal Circulation Research.
The new method allows ablation procedures to be tailored to the specific needs of individual patients with persistent AFib, identifying the key regions to treat with high precision. Moreover, the method costs no more than the conventional procedure and there is thus no obstacle to its use by most centers experienced in AFib ablation.
The investigators propose that the new algorithms could be easily incorporated into conventional electroanatomical navigation systems, increasing the precision and reducing the cost of patient-specific procedures for the ablation of persistent AFib.
Quintanilla, J. G., Alfonso-Almazan, J. M., Perez-Castellano, N., Pandit, S. V., Jalife, J., Perez-Villacastin, J., & Filgueiras-Rama, D. (2019). Instantaneous Amplitude and Frequency Modulations Detect The Footprint of Rotational Activity and Reveal Stable Driver Regions as Targets for Persistent Atrial Fibrillation Ablation. Circulation Research, 125(6), 609-627. doi:10.1161/CIRCRESAHA.119.314930
JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
The rate of coronary intervention in coronary-syndrome patients: an index of health system performance linked to survival
CNIC scientists have found that that a higher rate of coronary revascularization during hospitalization for non-ST segment elevation acute coronary syndrome (NSTEACS) is associated with better patient survival 2 years after hospital discharge, whether analyzed at the hospital, national, or supranational level.
The article was published in the Journal of the American College of Cardiology, and CNIC researchers Héctor Bueno, Xavier Rosselló, and Stuart Pocock believe that the findings will help to define and update quality-care guidelines for acute myocardial infarction, such as those recommended by the Acute Cardiovascular Care Association of the European Society of Cardiology. The revascularization rate in patients admitted for NSTEACS can serve as an index of health-care quality at the hospital, national, or supranational level. The CNIC researchers also affirmed that these results highlight “the importance of addressing the mismatch between clinical practice guideline recommendations and common medical practice.”
One of the study’s main conclusions is that there is a need for more dynamic referral procedures to ensure that NSTEMI patients are rapidly transferred from centers with limited facilities to centers with a catheterization laboratory.
Bueno, H., Rossello, X., Pocock, S. J., Van de Werf, F., Chin, C. T., Danchin, N., Lee, S. W., Medina, J., & Huo, Y. (2019). In-Hospital Coronary Revascularization Rates and Post-Discharge Mortality Risk in Non-ST-Segment Elevation Acute Coronary Syndrome. Journal of the American College of Cardiology, 74(11), 1454-1461. doi:10.1016/j.jacc.2019.06.068
Cell Metabolism
A new mechanism for the transfer
of maternal genetic material
CNIC researchers have defined the dynamics of the transfer of mitochondrial DNA, a type of genetic material, from mothers to their offspring. The study was published in Cell Metabolism. Using experimental mouse models, the research team led by Dr José Antonio Enríquez found that when the mother’s cells contain more than one mitochondrial DNA variant, this genetic transfer from mother to pup is controlled by two mechanisms: first during the development of the egg (oocyte), and later during the early stages of embryonic development. The scientists explained that “this control is aimed at preventing the co-occurrence of various types of mitochondrial DNA in the new individual,” which can lead to the development of mitochondrial diseases.
The information provided in the new study is important for two reasons. Understanding the mechanisms that regulate the segregation of the mitochondrial genome is necessary for the development of strategies to prevent mother-to-child transfer of mutated mitochondrial DNA that cause mitochondrial diseases. The new findings will also help scientists to devise ways to prevent different types of mitochondrial DNA co-occurring in the same cell as an unwanted result of medical interventions; this phenomenon, known as heteroplasmy, is a potential risk of mitochondrial replacement therapy.

The study shows that heteroplasmy can alter the metabolism of embryonic cells, inducing increased mitochondrial production of reactive oxygen species. This produces changes in the morphology of the inner mitochondrial membrane and in the molecular machinery for energy production.
Latorre-Pellicer, A., Lechuga-Vieco, A. V., Johnston, I. G., Hamalainen, R. H., Pellico, J., Justo-Mendez, R., Fernandez-Toro, J. M., Claveria, C., Guaras, A., Sierra, R., Llop, J., Torres, M., Criado, L. M., Suomalainen, A., Jones, N. S., Ruiz-Cabello, J., & Enriquez, J. A. (2019). Regulation of Mother-to-Offspring Transmission of mtDNA Heteroplasmy. Cell Metabolism, 30(6), 1120-1130. doi:10.1016/j.cmet.2019.09.007
JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
An oral anticoagulant delays the appearance of Alzheimer disease in mice
Scientists at the CNIC have identified a possible treatment for Alzheimer disease. Working together with a scientific team at The Rockefeller University in New York, the investigators have shown that treatment with the oral anticoagulant dabigatran delays the appearance of Alzheimer disease in mice.
The results, published in the Journal of the American College of Cardiology, show that after a year of treatment with dabigatran, mice had no memory loss and no reduction in cerebral circulation. This treatment also reduced typical Alzheimer symptoms, including cerebral inflammation, blood vessel injury, and amyloid protein plaques.

Dabigatran is more effective, has fewer side effects than classical anticoagulants, and is approved for the treatment of several diseases. Study coordinator Dr Marta Cortés Canteli, a CNIC researcher funded through the Miguel Servet program, highlighted the value of the study: “This discovery marks an important advance toward the translation of our results to clinical practice to achieve an effective treatment for Alzheimer disease.”
Placing the study in a broader context, Dr Valentín Fuster, CNIC General Director and a lead author on the study, commented, “neurodegenerative diseases are very closely linked to disease in the cerebral blood vessels. The study of the links between the brain and heart is the major challenge for the next ten years.”
Further studies are needed in patients, but the research team believe that dabigatran has the potential to normalize cerebral blood flow in Alzheimer patients. “An individualized treatment strategy such as this will first require the development of a diagnostic tool to identify those Alzheimer patients with a tendency to coagulation. This will be an important line of research in the coming years,” indicated Dr Cortés Canteli.
Cortes-Canteli, M., Kruyer, A., Fernandez-Nueda, I., Marcos-Diaz, A., Ceron, C., Richards, A. T., Jno-Charles, O. C., Rodriguez, I., Callejas, S., Norris, E. H., Sanchez-Gonzalez, J., Ruiz-Cabello, J., Ibanez, B., Strickland, S., & Fuster, V. (2019). Long-Term Dabigatran Treatment Delays Alzheimer’s Disease Pathogenesis in the TgCRND8 Mouse Model. Journal of the American College of Cardiology, 74(15), 1910-1923. doi:10.1016/j.jacc.2019.07.081
Nature Communications
The ‘airbag’ that protects cells against stress
Scientists at the CNIC have identified the molecular mechanisms that allow our cells to adapt to, protect themselves against, and survive mechanical stress. The results, published in Nature Communications, show that our cells produce molecules that act as a type of ‘airbag’ in response to mechanical stress. Without this protective and adaptive system, the heart, an organ subject to continuous mechanical forces, “would be unable to correctly perform its blood-pumping role,” explained lead author Miguel Ángel del Pozo. First author Asier Echarri added that the findings “show the importance of identifying the molecular mechanisms that protect cells against mechanical stress.”
The findings are important because knowledge about how cells are protected against mechanical stress “will give us a better understanding of the molecular basis of diseases such as some forms of muscular dystrophy, cardiomyopathies, and lung or vascular diseases characterized by sensitivity to physical activity. The findings will also shed light on the mechanisms of injury to organs with a high level of mechanical activity, such as the heart, lungs, muscles, and blood vessels.”
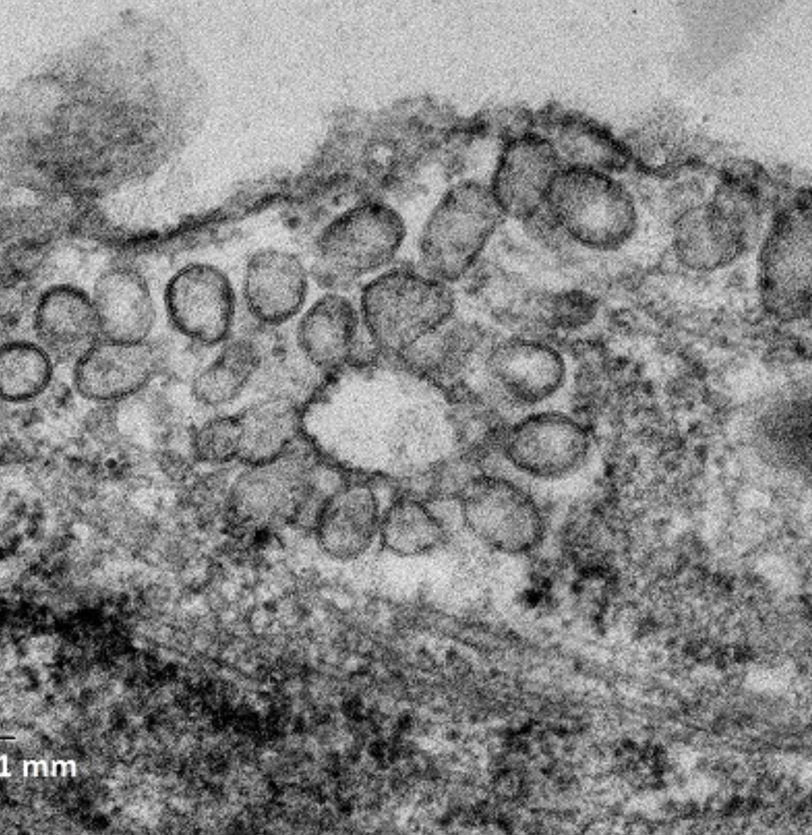
Echarri, A., Pavon, D. M., Sanchez, S., Garcia-Garcia, M., Calvo, E., Huerta-Lopez, C., Velazquez-Carreras, D., Viaris de Lesegno, C., Ariotti, N., Lazaro-Carrillo, A., Strippoli, R., De Sancho, D., Alegre-Cebollada, J., Lamaze, C., Parton, R. G., & Del Pozo, M. A. (2019). An Abl-FBP17 mechanosensing system couples local plasma membrane curvature and stress fiber remodeling during mechanoadaptation. Nature Communications, 10(1), 5828. doi:10.1038/s41467-019-13782-2
4. CNIC News and Views
1. GRANTS
REANIMA
toward a new paradigm in cardiac regeneration
Research into new endogenous mechanisms of tissue regeneration is an innovative research avenue in cardiac regeneration. This is the central goal of the REANIMA project (New‐generation cardiac therapeutic strategies directed to the activation of endogenous regenerative mechanisms), a research program coordinated by Dr Miguel Torres at the CNIC and supported by €8 million funding over 5 years, of which €1,380,000 is directly managed at the CNIC. The project started in January 2020.
The project brings together knowledge obtained from species that can regenerate their hearts (fish and amphibians), animals that cannot (adult mammals), and human heart tissues generated by tissue engineering.
REANIMA is the first Europe-wide project to integrate basic research findings in an effort to transform them into medical applications in the field of cardiac regeneration. REANIMA is funded by the European Union’s Horizon 2020 research and innovation programme under grant agreement nº 874764. Project activities range from identifying new targets in animal models to the design of clinical trials. The project is linked to the similarly named ‘REANIMA-CM’ project funded by the Comunidad de Madrid, which is also coordinated by Dr Torres.
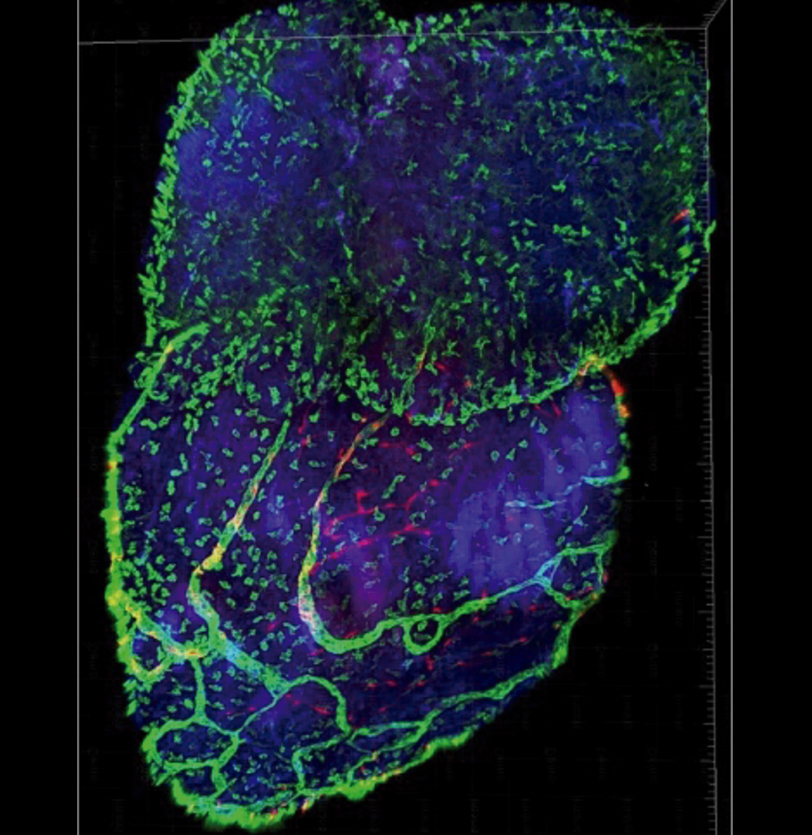
By bringing together industrial partners and academics specialized in translational and preclinical research, REANIMA will allow the development of new advanced therapies.
In addition to the CNIC, another 11 European research centers are participating in this innovative project.
REANIMA was selected for funding in the “Regenerative medicine: from new insights to new applications” call, published within the “Health demographic change and wellbeing” H2020 Work Programme. This call had a total Budget of €50,000,000 and received 154 project submissions. REANIMA was the second ranked project and received the maximum permitted funding, a total of €8 million for 12 European institutions in several countries and covering a period of 5 years.
NeutroCure
exploiting the dual nature of neutrophils to cure inflammatory diseases
Curing disease with neutrophils; this is the goal of the NeutroCure project. How will that work? “Neutrophils have a yin yang character,” explains Andrés Hidalgo, whose team at CNIC is one of six member groups participating in this project coordinated by Andriy Mokhir Friedrich at Alexander University, Germany.
NeutroCure focuses on reactive oxygen species (ROS) produced by neutrophils. In healthy organisms, ROS play crucial roles, such as signaling to regulate cell growth, providing the trigger for the formation of neutrophil extracellular traps, and modulating inflammation. However, high ROS concentrations damage tissues, and nature has therefore evolved precise mechanisms to control ROS duration and concentration and to ensure that these molecules remain confined to locations close to their targets.
Disruption of these mechanisms causes aberrant ROS production, leading to uncontrolled inflammation. This occurs, for example, during myeloablation triggered by radiotherapy or chemotherapy, and is a key characteristic of the phenotype of cancer cells and autoimmunity.
The goal of NeutroCure is to ensure that these substances act selectively and are controlled. To achieve this, the consortium members focus on the protein elastase, which is specific to neutrophils. The team has designed a ‘chemical cage’ that opens to ‘release’ ROS only when elastase is in highly defined anatomical locations and contexts.
The project will assess this approach in models of inflammatory disease, including arthritis, multiple sclerosis, autoimmune neuritis, lupus, and psoriasis.
The project has received European funding of €3 million for 5 years in the category H2020-FETOPEN-2018-2020 - FET Open – Novel ideas for radically new technologies. The CNIC team led by Andrés Hidalgo will receive €400,000 as one of the six academic groups in the NeutroCure consortium.
The NeutroCure consortium also includes a private company, which will steer the commercialization of new medication developed in the project. The consortium members anticipate that NeutroCure will have a major societal impact by providing new treatments for severe disorders caused by the dysregulated production of ROS.
EXPLOSIA
2 M euros from the European Research Council for a ground-breaking research project on atherosclerosis
Professor Jacob Fog Bentzon of Aarhus University and the CNIC has received €2 million from the European Research Council for a ground-breaking research project on atherosclerosis, one of the most frequent causes of death in the world.
Although atherosclerosis is a very common disease, much work remains to be done to define exactly what happens when fat and cholesterol are deposited in the walls of arteries, which leads to arteriosclerosis and the risk of blood clots in the heart and the brain.
Scientists only recently discovered that many of the cells found in atherosclerotic plaques are in fact vascular smooth muscle cells that have altered their appearance and function. The research group led by Professor Fog Bentzon will use the new funding to uncover the significance of these mysterious cells.
“When you look at atherosclerosis under the microscope, you see a complex mix of many types of cells. But we now know that although the cells may look different, most of them originate from the arteries’ own vascular smooth muscle cells,” says Bentzon.
“Our ultimate goal is to find new ways to treat advanced atherosclerosis in humans. And the route to this goal passes through a wide range of studies in genetically modified mice and pigs,” he adds.
The Fundación Bancaria La Caixa selects 2 CNIC projects in its “Proyectos de Investigación en Salud 2018” program
Two CNIC projects were selected by the Fundación Bancaria La Caixa for its Proyectos de Investigación en Salud program for 2018.
The projects are ‘Dysfunction of Ion Channel Complexes in Inheritable Cardiac Diseases’, led by Dr José Jalife, and ‘Nitric Oxide signaling and proteoglycans in Marfan syndrome aortopathy: mechanisms and new therapeutic targets’, led by Dr Juan Miguel Redondo.
Dr Jalife’s project adopts pioneering approaches to define the causes of sudden cardiac death in inherited cardiac diseases and identify new and more effective prevention strategies. The new techniques employed in the study include the injection of adeno-associated virus to target mutated genes to the heart cells of mice, the derivation of cardiomyocytes from human induced pluripotent stem cells, and advanced proteomics and bioinformatics approaches. The project was awarded €499,895.

Dr Redondo’s project investigates the pathophysiological mechanisms underlying Marfan syndrome and the formation of aortic aneurysms and dissections. The goal is to identify new molecular targets and biomarkers that can be used to develop effective treatments. The project was awarded €500,000.
CNIC researcher secures funding for 2 projects from the European Federation for the Study of Diabetes
CNIC postdoctoral researcher Ivana Nikolić has been awarded funding for 2 projects by the European Foundation for the Study of Diabetes (EFSD) to study the role of T cells in the development of diseases associated with obesity. The 2 awards are the EFSD–Lilly Young Investigator Award, providing €50,000 and the EFSD Rising Star Fellowship, bringing another €30,000. Dr Niković previously obtained the 2017 EFSD–Lilly Research Fellowship Award, receiving a grant of €50,000.
Dr Nikolić obtained her PhD in Immunology at the University of Belgrade in 2014. In 2015 she joined Dr Guadalupe Sabio’s lab at the CNIC with an FP7-People Marie Sklodowka - Curie Actions, MSCA - COFUND talent recruitment fellowship. These fellowships were intended to attract outstanding foreign scientists to the CNIC at an intermediate stage in their career so that they could develop their research in Spain.

Dr Nikolić exemplifies the success of this talent recruitment program. The 3 EFSD awards she has obtained support her research into the molecular mechanisms of obesity.
2.AWARDS & HONORS
Valentín Fuster awarded the 2019 National Research Prize
The Minstry of Science, Innovation and Universities awarded the 2019 Gregorio Marañon National Research Prize to Valentín Fuster. The jury panel awarded the prize to Dr Fuster in recognition of his unparalleled research record and his enormous contributions to the prevention, diagnosis, and treatment of cardiovascular disease. The National Research Prize, first awarded in 1982, is the highest Spanish accolade for scientific research. The aim of the award is to reward the achievements of Spanish scientists who are international leaders in their research fields and who make exceptional contributions to the advance of scientific knowledge, technology transfer, and human progress.

Pura Muñoz-Cánoves awarded the Jaume I Prize for Medical Research

CNIC group leader Dr Pura Muñoz-Cánoves was awarded the XXXI Rei Jaime I prize in the Medical Research category. The Rei Jaume I are awarded to people who have made outstanding contributions in their fields through work carried out mostly in Spain. The prizes are awarded annually, and each prize winner receives a gold medal, a diploma, and €100,000, on the condition that part of this money be used to fund research and enterprise in Spain.
The prize was awarded to Dr Muñoz for her work on the molecular mechanisms of aging, especially the progressive changes that take place in the stem cells of animals as they age and the possibility of manipulating these changes for regenerative medicine.
Dr Muñoz was also awarded the Lilly Foundation Preclinical Biomedical Research Prize for 2019 in recognition of her pioneering work on tissue regeneration.
Rodrigo Fernández receives the Marie Skłodowska-Curie Actions Award
CNIC researcher Dr Rodrigo Fernández Jiménez received the 2019 Marie Skłodowska-Curie Actions Award in the ‘Scientific Careers for Policymaking’ category for his Comprehensive Lifestyle Intervention Project (CLIP).
CLIP investigates new approaches to health promotion and disease prevention. The goal is to help policy makers devise effective strategies to promote a healthy life style among children.
The aim of the project is to convince governments that low-cost and side-effect–free health-promotion programs aimed at young people provide a potentially effective way to reduce the cardiovascular disease epidemic if integrated into national school curriculums.

Sara Cogliati selected for a XIV L’Oréal-Unesco Research Prize For Women in Science

CNIC researcher Dr Sara Cogliati was awarded one of 5 prizes in the XIV edition of L’Oréal-Unesco For Women in Science Program for her project investigating sex-specific characteristics in cardiovascular disease. This research is crucial to finding effective treatments for heart failure in women because historically most treatments have been based on research in men, despite important differences between the sexes in the way the disease manifests. Dr Cogliati’s research signals an important advance toward gender equality in the treatment of disease.
Miguel Torres elected a member of the European Molecular Biology Organization
CNIC group leader Dr Miguel Torres has been elected a member of the European Molecular Biology Organization (EMBO). In the words of EMBO Director Maria Leptin, “EMBO members are expert scientists who carry out pioneering research across all disciplines in the life sciences, from computer models or analysis of individual molecules and cell mechanisms to the study of higher-level systems in development, cognitive neuroscience, and evolution.”
Dr Miguel Torres’s research focuses on the regulation of embryonic development and the formation and regeneration of organs. He has made major contributions to the understanding of how gene activities regulate regionalization processes in the developing embryo and the discovery of mechanisms involved in quality control and organ regeneration.

Among his various projects, Dr Torres directs the first Spanish-coordinated international project funded by the prestigious Leducq Foundation. The project—Redox Regulation of Cardiomyocyte Renewal—aims to identify how and why adult mammals lose the capacity to regenerate the heart after injury, whereas this ability is retained in newborn infants and in adults of other animal species, including fish and amphibians.
Francisco Sánchez-Madrid awarded an honorary doctorate (Doctor Honoris Causa) by the University of Córdoba

Professor Francisco Sánchez Madrid was awarded an honorary doctoral degree by the University of Córdoba. Dr Sánchez Madrid heads the Intercellular Communication in the Inflammatory Response laboratory at the CNIC and is full professor in Immunology at Madrid Autonomous University and Scientific Director of the Research Institute and Hospital de La Princesa, Madrid.
3. SCIENTIFIC EVENTS
CNIC Conference
new concepts in age-related cardiovascular disease
The CNIC held its annual scientific meeting, the CNIC Conference, on October 24 to 26. The 2019 conference, ‘New concepts in age-related cardiovascular disease’, was organized by three CNIC researchers, Vicente Andrés, José J Fuster, and Andrés Hidalgo, and Columbia University professor Allan Tall. The meeting hosted world experts in the field of aging related to cardiovascular disease (CVD).
Population aging is one of the most important demographic phenomena of our time, and its global impact is medical, social, and economic. Age is also the most important risk factor for CVD, yet the underlying mechanisms linking aging and CVD are still not fully understood. In recent years, studies have been published on a series of mechanisms specifically linked to aging that are completely independent from traditional risk factors. These new mechanisms, which are relevant to both cardiovascular and cerebrovascular disease, open the door to new therapies and prevention strategies.

Renowned participants included CNIC Director Dr Valentín Fuster, Dr. Filip Swirski from Harvard University, Costantino Iadecola from the Cornell School of Medicine in New York, and Paul Frenette from the Albert Einstein College of Medicine, who delivered the Company of Biologists keynote presentation. Leading Spanish experts in aging and cerebrovascular diseases attending the meeting included Dr M Ángeles Moro from Madrid Complutense University and Dr Manuel Serrano from the Biomedical Research Institute in Barcelona, who presented the EMBO keynote presentation.
The CNIC and CNIO joint workshop to share knowledge and boost synergies
Cancer and cardiovascular disease share many of the same molecular mechanisms, and it is common for studies to involve participation from experts in both fields. This is never more true than in the case of the Centro Nacional de Investigaciones Oncológicas (CNIO) and the Centro Nacional de Investigaciones Cardiovasculares (CNIC), which boast similar and complementary technological platforms. These neighboring centers of excellence, both under the umbrella of the Instituto de Salud Carlos III (ISCIII), organized the CNIO–CNIC Joint Meeting to share knowledge, boost synergies, and promote joint projects.
The meeting was overseen by ISCIII Director Raquel Yotti, CNIO Director Maria Blasco, and CNIC Director Valentín Fuster. Participants reviewed the most advanced research lines at each center and emphasized the vital importance of collaborative research.
CNIC PhDay 2019
Since its launch in 2014, the annual PhDay event has provided a platform for proposing and debating diverse science-related topics of interest to early-stage researchers. The main goals of the event are to help participants develop their careers as scientists, exchange new ideas, and establish career networks. This year's event had more than 200 participants, and the chosen theme, 'What else?', centered on career options. Entrepreneurs and scientists from academia and industry joined to discuss life after a PhD and the different paths available.

Invited CNIC Seminars
During 2019, the CNIC hosted 23 invited speakers, who delivered presentations at the Center.
4. OUTREACH ACTIVITIES
SCIENCE WEEK AT THE CNIC
For Semana de la Ciencia 2019, the CNIC introduced 2 new activities. The ‘Family Day at the CNIC’ event was designed especially for younger children and their families, while the #ConCienciaCNIC one-day events consisted of scientific workshops for children with special needs.
These activities catered for children between the ages of 4 and 14, accompanied by adults, and aimed to introduce tomorrow’s scientists to the world of research. Through theater, play activities, and scientific workshops, the children had fun, but also learned from the experiments carried out by CNIC volunteers.

5.NEW PARTNERSHIPS
The CNIC and the SEC launch a study aimed at changing clinical practice after an infarctionafter an infarction
The CNIC and the Spanish Society of Cardiology (SEC) joined forces to launch a pioneering clinical trial in Spain: TREatment with Beta-blockers after myOcardial infarction withOut reduced ejection fracTion (REBOOT). In the words of prinicipal investigator Dr Borja Ibáñez, “the ambitious aim of this trial is to change clinical practice guidelines for the treatment of acute myocardial infarction.” The trial will test the effect of maintaining beta-blocker therapy after hospital discharge in a cohort of 8500 patients.
Beta-blockers are drugs that reduce heart rate, blood pressure, and cardiac contractility (strength of contraction). This promotes cardiac diastole (filling), which improves heart function and blood flow in the coronary arteries. Most of the evidence supporting the use of beta-blockers in heart attack patients comes from before the current era of widespread use of reperfusion therapy. Despite this, the recommendations in European and American guidelines for beta-blocker therapy in these patients have remained unchanged for decades.
REBOOT will be the first trial in the reperfusion era to study if the administration of beta-blockers after an acute myocardial infarction influences the incidence of death, reinfarction, and hospital readmission for heart failure.
The trial will randomize 8500 patients to receive beta-blocker therapy or no beta-blocker therapy. Participants will be followed up for a minimum of 2 years and a maximum of 3 years. Outcome measures will include the incidence of clinical events and adherence to the randomized therapy, recorded at 13, 15, and 36 months. A subsample of 1000 patients will be assessed for quality of life during follow-up.
This research topic is of such high relevance that another 3 European trials similar to REBOOT are planned, in Sweden, Norway, and Denmark. In total, more than 20,000 patients with similar characteristics will be randomized to beta-blocker therapy or no beta-blocker therapy after a myocardial infarction with ventricular dysfunction.
6.SOCIAL & CNIC
The Pro-CNIC Foundation renews its commitment to the CNIC until 2028
Pro-CNIC Foundation President Luis de Carlos and General Secretary of Scientific Coordination Rafael Rodrigo formalized an agreement to renew the commitment of this body of 12 private companies to support the CNIC until 2028. The Pro-CNIC Foundation is an innovative example of public-private collaboration that has allowed the CNIC to establish itself as a world leader in biomedical research, contributing to the CNIC gaining Severo Ochoa accreditation in recognition of its international research excellence. Through the Pro-CNIC Foundation, 12 leading Spanish companies channel their business knowhow and financial support to the CNIC, representing a benchmark in successful scientific patronage. The companies participating in the Pro-CNIC Foundation are Acciona, Santander Bank, BBVA Bank, Endesa, the Mapfre Foundation, the Mutua Madrileña Foundation, the Ramón Areces Foundation, the Repsol Foundation, Inditex, “la Caixa”, Prisa, and Telefonica.
The Pro-CNIC Foundation enables the CNIC’s research to have a direct impact on patient care and health improvements in the general population. The Pro-CNIC Foundation also helps in the conversion of innovations into patents that generate an economic return and support research and development in Spain.

A heart attack survivor donates book sale proceeds to the CNIC
A heart attack survivor is donating the proceeds from her book to the CNIC after ‘coming back to life’. On July 14, 2014, Paula Elena Ramos came back from the doors of death. Without warning, and with no family history or prior symptoms, Paula Elena had a third-degree atrioventricular block. She tells her story in her book a“Historia del corazón que sembró una galaxia en un cuerpo eléctrico,” and all income from the sale of the book are being donated to arrhythmia research at the CNIC, specifically in the Advanced Development in Arrhythmia Mechanisms and Therapy laboratory led by Dr. David Filgueiras.

Participants in the V Congress of Scientist-Entrepreneurs visit the CNIC

AAs part of the activities program linked to the V Congress of Scientist-Entrepreneurs in May, a group of participants spent a day at the CNIC learning about the center’s activities and installations.
The visited facilities included the Advanced Infrastructure for Translational Imaging (TRIMA), part of the ICTS Distributed Biomedical Imaging Network (ReDIB).
5. Training Programs
Training programs and continuing education

Training is one of the CNIC’s core activities, and the Center has devised a comprehensive training plan, the CNIC-Joven Training Plan. This global plan includes programs for participants at all levels, from high-school students to postdoctoral researchers and MDs. The CNIC-Joven Training Plan aims to fulfill the personal goal of Valentín Fuster “to attract and train the brightest young people from the earliest ages to create a pool of excellent researchers in the field of cardiovascular research.”
In 2019, 473 participants were enrolled on CNIC training programs.
HIGH SCHOOL a STUDENTS INTERNSHIPS
Acércate Program
The Acércate Program offers the highest-achieving senior high school students in the natural and health sciences the chance to experience life as a biomedical researcher, with the aim of awakening and strengthening interest in a biomedical research career.
Participants spend two weeks at the CNIC, learning modern techniques used in biomedical research, conducting supervised experiments, operating sophisticated scientific equipment and presenting the results of their work, all under the supervision of CNIC researchers.
Fellowships in 2019: 8
The CNIC collaborates with high schools of the Madrid Community in the following training programs:
4ºESO-CNIC: Sixteen life-science students from eight schools spent from 4 to 5 days at the CNIC laboratories exploring possible scientific careers.
The CNIC was also visited by 12 year-4 high-school students as part of the Programa 4ºESO+CNIC 2019. This program provides educational visits to businesses and observation sessions in a professional setting to students in their final year of compulsory secondary education. The aim is to promote a vocation for science among young students. The 12 students visiting the CNIC came from 7 high schools: IES Fortuny, IES Santa Teresa de Jesús, Colegio Raimundo Lulio, and Colegio Santa Francisca Javier Cabrini in Madrid and Colegio Humanitas de Torrejón de Ardoz, Colegio Gredos San Diego El Escorial, and Colegio Calasanz in Alcalá de Henares.

Practical experience for Technical School students: This program brought eighteen technical school students studying “Pathological and Cytodiagnostic Anatomy” and “Clinical and Biomedical Laboratory Science” to gain practical curricular experience in the CNIC’s laboratorios over a three-month period. Moreover, three students studying “Diagnostic Imaging and Nuclear Medicine” and “Clinical and Biomedical Laboratory Science” spent 10 months at the CNIC as part of the DUAL technical study program. The CNIC has signed collaborative agreements for this kind of interships with both DUAL Centers in Madrid offering courses in the Biomedicine field.

PROGRAMS FOR UNDERGRADUATE STUDENTS
Internships are offered to university students in the following programs:
Cicerone Program
The Cicerone Program is open to advanced undergraduate students and Master’s students in biomedicine-related disciplines. Participants extend their scientific training through hands-on experience of laboratory-based biomedical research during the summer recess. In addition to carrying out a supervised research project, the students also attend CNIC seminars and workshops. The aim of the program is to give students first-hand knowledge of biomedical research so that they can make informed choices about the possibility of pursuing a scientific career.
Fellowships in 2019: 29
Curricular and Extracurricular
University Practical Program
The CNIC offers practical training in cardiovascular research to visiting undergraduate students, including those on Erasmus internships.
Internships in 2019: 70 internships from 12 Universities, 4 of them outside Spain.
MASTERS AND GRADUATE STUDENTS
Master’s Fellowship Program (CNIC-ACCIONA) and Fundacion Carolina BBVA-CNIC Master’s Fellowship Program
These grants provide funding for students studying for a master’s degree at a Spanish university to conduct their experimental project (TFM) in a CNIC laboratory.
Fellowships in 2019: 15
Predoctoral (PhD) Program
The Predoctoral Program provides a unified framework for all CNIC researchers who are working toward a doctoral degree. All predoctoral researchers are signed up to this program, irrespective of their funding source.
The aims of the program are to ensure uniform quality of predoctoral training at the CNIC and further to ensure fair and equal access of predoctoral researchers to training opportunities.
The Program schedules regular meetings between the predoctoral fellow and his or her thesis committee, composed of the thesis director, another CNIC group leader, and an external expert.
Graduate students at the CNIC awarded a PhD degree in 2019: 17
Graduate students studying for a PhD degree at the CNIC in 2019: 100

Insights into Research in Cardiovascular Disease Masters Module
This postgraduate course is run by the CNIC as part of the Universidad Autónoma de Madrid (UAM) Molecular Biosciences Master’s Program. This optional module provides a broad overview of cardiovascular biology, including perspectives from basic, clinical, and translational research.
Course participants are enrolled UAM Master’s students, CNIC predoctoral researchers, and participants on the Res@CNIC SEC Program (see below).

MEDICAL INTERN RESIDENTS
RES@CNIC Program
The Res@CNIC-SEC Program (in collaboration with the Spanish Society of Cardiology, SEC) offers resident medical interns the opportunity during the first years of their specialization period to learn about the latest techniques in cardiovascular research being used in the CNIC’s laboratories, under the guidance of a CNIC scientist. Residents participating in RES@CNIC also receive training in theoretical aspects of cardiovascular research through an expert-led taught module. The Program also seeks to create links and collaborations so that on conclusion of their MIR specialization period, these professionals will have the chance to undertake research projects in their respective Hospitals in partnership with CNIC scientists.
Participants in 2019: 19
INVESMIR SEC Program
The INVESMIR SEC Program offers resident medical interns the opportunity during their specialization period to further their training through a research project in one of the CNIC’s laboratories, under the supervision of a CNIC scientist.
An important aim of the program is for participants to establish contacts and collaborations with CNIC researchers that will support them, after completion of their MIR specialization training, in pursuing their own research projects at their centers within the Spanish National Health System. In 2019 two resident cardiologist interns participated in this program.
RESEARCHERS
Cardio Joven Program
The aim of this program (also organized in collaboration with the SEC) is to foster high-quality translational research in the cardiovascular area in Spanish National Health System centers through training programs providing theory and practical training for cardiologists with a research vocation.
Specific aims:
a) To create the figure of the cardiologist-researcher by providing high-quality training in clinical research methods, including statistical analysis and the latest basic research techniques used in cardiovascular biomedicine, as well as opportunities to explore any clinical area of cardiology in greater depth (sub-specialization).
The program is aimed at cardiologists who aspire to carry out advanced clinical and research work at any center within the Spanish National Health System.
b) International training. The program offers a period of training toward a Master’s Degree in Epidemiology at the London School of Hygiene and Tropical Medicine (90 ECTS)
Post MIR SEA CNIC Program
This program offers 1 -or 2- year contract for research into electrophysiology and arrhythmias. This contract is available to a physician completing their resident intern specialization (MIR) in cardiology and to members of the SEC Sección de Electrofisiología y Arritmias.
CONTINUING TRAINING
Cardiovascular Pathophysiology Course: From Symptoms to Genes
The two-day Cardiovascular Pathophysiology Course is organized by the CNIC (Borja Ibáñez) in partnership with the SEC (Julián Pérez Villacastín). Participants receive an overview of the molecular and genetic factors that underlie cardiac diseases and gain an up-to-date vision of cardiac physiology.
Venue: CNIC Auditorium
Dates: December, 13-14, 2019
Attendees: 91
Vascular Biology Course
Valentín Fuster delivers this lecture series on Vascular Biology: Basic and Clinical Research as part of the Universidad Internacional Menéndez Pelayo (cUIMPB) summer program, sponsored by Ferrer International.
In this course, Valentín Fuster tries to “motivate and teach for the future”. Most of the attendees are cardiologists, although others are experts in internal medicine or other specialties. In 2018, delegates came from more than 15 countries, most of them in Latin America and Europe.
Venue: Cardona Auditorium
Dates: July, 15-16, 2019
Attendees: 100

6. Facts and Figures
Scientific Publications
| DOCUMENTS | N. | |
|---|---|---|
| Articles | 211 | 80% |
| Reviews | 23 | 9% |
| Other | 29 | 11% |
| TOTAL | 263 |
LEADERSHIP
56%
CNIC-Led
publications
OPEN ACCESS
44%
| DOCUMENTS | |
|---|---|
| D1 | 45% |
| Q1 | 79% |
| Q2 | 17% |
Publications
in Top
Journals
(IF>10)
7930%
| AFFILIATION | ||
|---|---|---|
| CNIC | 18 | 7% |
| National Collaboration | 76 | 29% |
| International Collaboration | 34 | 13% |
| National and International COLLABORATION | 135 | 51 |
Collaboration
with Hospitals
69%
Collaboration
with Universities
88%
Competitive Funding
 NEW GRANTS
NEW GRANTS

| National | International |
|---|---|
| 71 Grants | 13 Grants |
| €8,160,000 | €2,860,000 |
Technology Transfer
| 6 | Patent applications |
|---|---|
| 5 | Licensed patents |
| 7 | Confidential Disclosure Agreements |
| 1 | New collaboration agreement with industry |
| 78 | Material Transfer Agreements |
HUMAN RESOURCES
Scientific Staff
415
-

-
women 253 162 MAN
Research Areas
348
Group Leaders
31
-

-
women 8 23 MAN
Technical Units
67
Cardiologists
7
Heads of Technical Units
11
-

-
women 5 6 MAN
42(10%)
of the Scientific Staff are from outside Spain
Visiting Scientists
240
-
-
(from institutions from outside Spain 37 – 17 countries including Switzerland, Denmark, Germany, Italy, UK, and USA)
7. Acknowledgments
BOARD OF TRUSTEES
Pedro Francisco Duque Duque
Minister of Science and InnovationRafael Rodrigo Montero
General Secretary for Scientific Policy Coordination Ministry of Science, Innovation and UniversitiesRaquel Yotti Álvarez
Director, Instituto de Salud Carlos IIIAPPOINTED MEMBERS
Faustino Blanco González
General Secretary for Health and Consumers AffairsMinistry Health, Consumers Affairs and and Social Welfare
Rosa Menéndez López
President of the State Agency of the Spanish National Research Council (CSIC)Ministry of Science, Innovation and Universities
Margarita Blázquez Herranz
Deputy Director General of Networks and Cooperative Research Centers, Instituto de Salud Carlos IIIEnrique Ruiz Escudero
Health Counselor of the Madrid RegionCarlos Macaya Miguel
President of the Spanish Heart FoundationELECTED MEMBERS
Luis de Carlos Bertrán
President of the ProCNIC Foundation BoardFundación Mapfre
Julio Domingo Souto General DirectorBanco Santander
Rodrigo Echenique Gordillo Vicepresident, Banco SantanderTelefónica, S.A.
Francisco de Bergia González Director of Public Affairs of the PresidencyDeputy: Loreto Pérez del Puerto Rodríguez Public Affairs of the Presidency
Fundación Bancaria “la Caixa”
Antonio Vila Bertrán General DirectorDeputy: Angel Font Vidal Public Affairs of the Presidency Corporate Director of Research and Strategy
GENERAL SECRETARY
Margarita Blázquez Herranz
Deputy Director General of Networks and Cooperative Research Centers, Instituto de Salud Carlos IIILEGAL ADVISER
Juan José Torres Fernández
State Law in the Supreme CourtDeputy: Fernando Carlos Fernández de Trocóniz Marcos
SCIENTIFIC ADVISORY BOARD
Karin R. Sipido
Division of Experimental Cardiology Katholieke Universiteit Leuven, BelgiumJavier Díez
GCentro de Investigación Médica Aplicada (CIMA) University of Navarra, Pamplona, SpainMEMBERS
Margaret Buckingham
Department of Developmental Biology Pasteur Institute, Paris, FranceAna Mª Cuervo
Albert Einstein College of Medicine New York, USAPeter Carmeliet
Center for Transgene Technology and Gene Therapy Flanders Interuniversity Institute for Biotechnology, Leuven, BelgiumElisabetta Dejana
Department of Biosciences University of Milan (IFOM-IEO Campus), ItalyStefanie Dimmeler
Institute of Cardiovascular Regeneration Centre for Molecular Medicine, Frankfurt, GermanyMauro Giacca
School of Cardiovascular Medicine & Sciences King’s College London, UKStephane Heymans
Department of Cardiology, Cardiovascular Research Institute Maastricht (CARIM), The Netherlands Center for Molecular and Vascular Biology, University of Leuven, BelgiumGerd Heusch
Institute of Pathophysiology, Center of Internal Medicine University of Essen Medical School Essen, GermanyPatrizio Lancellotti
University Hospital of Liège BelgiumJosé López-Barneo
Facultad de Medicina de la Universidad de Sevilla SpainFilip Swirski
Research Institute, Massachusetts General Hospital Boston, USADerek M. Yellon
University College London (UCL) London,UKPUBLIC AND PRIVATE BODIES OF COMPETITIVE GRANS
AMGEN
AUSTRIAN SCIENCE FUND
COMUNIDAD DE MADRID
EUROPEAN COMMISSION
EUROPEAN COOPERATION IN THE FIELD OF SCIENTIFIC AND TECHNICAL RESEARCH
EUROPEAN FEDERATION OF IMMUNOLOGICAL SOCIETIES
EUROPEAN FOUNDATION FOR THE STUDY OF DIABETES
EUROPEAN MOLECULAR BIOLOGY ORGANIZATION FOR THE STUDY OF DIABETES
EUROPEAN RESEARCH COUNCIL
FORD ESPAÑA APADRINA LA CIENCIA
FUNDACION BBVA
FUNDACION CIENTIFICA DE LA AECC
FUNDACION LA CAIXA
FUNDACION LA MARATO TV3
FUNDACION RAMON ARECES
HUMAN FRONTIER SCIENCE PROGRAM
INSTITUTO DE SALUD CARLOS III
LEDUCQ FUNDACION
MINISTERIO DE CIENCIA E INNOVACION
MINISTERIO DE UNIVERSIDADES
MUSCULAR DYSTROPHY ASSOCIATION
SOCIEDAD ESPAÑOLA DE CARDIOLOGIA
THE COMPANY OF BIOLOGISTS
THE MARFAN FOUNDATION
8. Figures legends and credits by Research Area
Vascular Pathophysiology
Figure 1- Transverse view of an E9.5 mutant heart with defective ventricular development that will cause embryonic death by E11.0. The myocardium in ventricles and atria is stained with an antibody against smooth muscle actin (red), and nuclei are stained with DAPI (blue). (José Luis de la Pompa)
Figure 2- PET/CT of gene-modified minipigs with atherosclerosis, showing the accumulation of the PET tracer sodium fluoride in atherosclerotic lesions in vivo. (Jacob Bentzon)
Figure 3- PET analysis of the tracer sodium fluoride in atherosclerotic lesions ex vivo. (Jacob Bentzon)
Figure 4- H&E staining of transverse section of an E16.5 mutant mouse heart carrying a mutation identified in pedigrees of a family with non-compaction cardiomyopathy. The mutant phenotype recapitulates the features of noncompaction. Note the thin ventricular walls and the persistent trabeculae that have not undergone compaction. (José Luis de la Pompa)
Figure 5- Decreased brain metabolism in middle-aged hypertensive individuals. 3D brain statistical parametric maps, with colored areas indicating the association between the presence of hypertension and reduced glucose uptake, independently of other cardiovascular risk factors. The correlation analysis included baseline plasma glucose as a covariate together with the following principal component factors: age, sex, systolic blood pressure, total cholesterol, HDL cholesterol, smoking, hypertension, and diabetes. The color bar represents the magnitude of voxel significance (P-value). Only clusters with k>100 statistically significant voxels were considered. L, left; R, right; A, anterior; P, posterior. (Marta Cortés Canteli, Valentin Fuster)
Figure 6- Confocal image of kidney (left) and liver (right) from Lmnaflox/flox SM22αCre mice, showing expression of lamin A/C (white), smooth muscle a-actin (red), CD31 (Green), and DAPI (blue). (Vicente Andrés)
Cell and Developmental Biology
Figure 1- Whole-mount imaging, 3D reconstruction, and electron microscopy of mouse heart tissue, with individual cardiomyocytes in red and surrounding cardiac macrophages in green. The images illustrate the close immune-cell–cardiomyocyte interactions in the heart, whose functional significance we are trying to understand. (Andrés Hidalgo)
Figure 2- siRNA "custom" collection dispensing service for High Content Screening in the Cellomics Unit. The Freedom EVO robotic pipetting platform (TECAN, Switzerland) performs automated liquid handling and cherrypicking from genome-wide source siRNA libraries, as well as custom siRNA plate spotting. (María Montoya).
Figure 3- Electron microscopy image of a cell plasma membrane, showing caveolar ´rossettes´. (Miguel Ángel del Pozo)
Figure 4- Cell-scale analysis of heart tube formation in an optical section of a mouse embryo at embryonic day E8.0 (ventral view). The image shows mesodermal cell membranes (green), dTomato-positive transgenic cells (red), and nuclei (DAPI, blue). The image was acquired with a Leica SP8 Navigator confocal microscope and deconvolved using Huygens Professional 19.10. The image resolution of the image allows for very precise quantitative studies at single-cell level. (Miguel Torres)
Figure 5- Artistic rendering of individual channels from the previous image – Fig. 4. (Miguel Torres)
Figure 6- Mouse embryo at gestation day E8.3. Cardiomyocytes (labeled with an antibody against α-Smooth Muscle Actin, red) form the linear heart tube at this stage. A nuclear reporter for Notch activity outlines the developing vasculature (green). Cell nuclei were counterstained with the nuclear marker DAPI (blue). (Miguel Torres)
Figure 7- Confocal microscopy image of adipocytes differentiated in vitro, stained for lipid droplets (BODIPY 493/503 stain, green), actin cytoskeleton (phalloidin, red), and nuclei (DAPI, blue). (Miguel Ángel del Pozo)
Myocardial Pathophysiology
Figure 1- Electron microscopy image of a lipid-filled peritoneal tissue resident macrophage. (Mercedes Ricote)
Figure 2- Excised heart of an LQT8 knockin pig ready for isolation of cardiomyocytes. (Silvia Priori)
Figure 3- Termination of persistent atrial fibrillation (AF) upon instantaneous amplitude modulation and instantaneous frequency modulation (iAM/iFM)-based guidance to localize specific atrial areas driving the overall arrhythmia. (David Filgueiras)
Figure 4- Characterization of cardiac macrophages in CX3CR1GFP/+ mice after myocardial infarction. The images show 30μm maximum intensity projections of cardiac macrophages labeled with GFP (green), CD68 (red), and DAPI (blue) at 3 (left) and 7 (right) days after injury. (Mercedes Ricote)
Figure 5- LQT8 human IPSC-derived myocytes Simultaneous recording of voltage and calcium. (Silvia Priori)
Figure 6- Confocal image of kidney (left) and liver (right) from Lmnaflox/flox SM22αCre mice, showing expression of lamin A/C (white), smooth muscle a-actin (red), CD31 (Green), and DAPI (blue). (Vicente Andrés)













This innovative public-private financing formula has allowed the CNIC to reach a very high level of excellence, as recognized in the Severo Ochoa accreditation and other international awards.
© Centro Nacional de Investigaciones Cardiovasculares (F.S.P.), 2020
DL: M-2575-2020