EHJ: Metoprolol: an old drug with unique cardioprotective properties
CNIC scientists have defined the unique ability if metoprolol to protect the heart during a heart attack
A drug costing less than 2 euros per dose could reduce the long term consequences of a heart attack, benefitting millions of patients. Metoprolol, a member of the beta-blocker class of drugs that has been in use for more than 40 years, has been found to have unique cardioprotective properties.
This is the conclusion of a study carried out by scientists at the Centro Nacional de Investigaciones Cardiovasculares (CNIC), Fundación Jiménez Díaz University Hospital, and the Cardiovascular Disease Research Network (CIBERCV). The study, performed in sophisticated experimental mouse models, shows that the cardioprotective effect of metoprolol during a heart attack is not shared by other beta-blockers commonly administered by intravenous injection, such as atenolol and propranolol.
The European Heart Journal study was led by Dr Borja Ibáñez, head of Clinical Research at the CNIC, a cardiologist at Fundación Jiménez Díaz University Hospital, and a CIBERCV group leader. The research “demonstrates that metoprolol has unique cardioprotective properties and heralds a paradigm change in cardiology and the treatment of acute myocardial infarction,” said Dr Ibañez.
In 2013, the METOCARD-CNIC clinical trial, led and coordinated by the CNIC, showed that administration of metoprolol very early during an infarction limits damage to the heart and reduces long term consequences.
Four years later, in 2017, the same research team showed how and why this cheap and simple therapeutic strategy is so efficient. In a study published in Nature Communications, the researchers showed that the cardioprotective potential of metoprolol lies in its ability to block the action neutrophils—inflammatory cells activated during infection to eliminate pathogens—preventing them from entering the infarcted heart tissue.
Now, in 2020, the team has shown that metoprolol’s cardioprotective properties are not shared by other beta-blockers and are thus not a class effect.
“The study presents important results that update and refine cardiovascular pharmacotherapy and underline how important it is not to assume that drugs in the same class will have identical activities and clinical indications,” said pharmacist Agustín Clemente, a doctoral candidate at the CNIC and first author on the article.
Acute myocardial infarction is one of the main manifestations of cardiovascular disease and is the leading cause of death in the world. In Spain, more than 70000 people have a heart attack every year. Current treatment guidelines recommend early administration of beta-blockers to patients with symptoms of an infarction but do not distinguish between the different drugs in this class. This is why the new results are so clinically significant.
The low cost of this drug, at less than 2 euros per acute treatment, adds value to this discovery
Neutrophils, as well as protecting against infection, can become hyperactivated in other situations, such as during a myocardial infarction, when they can cause significant additional injury to the heart. Metoprolol is able to limit this hyperactivation, thereby preventing inflammatory damage associated with infarction.
The study also assessed the effects of different beta-blockers in other models of inflammatory disease, like lung damage and peritonitis. In all models examined, metoprolol was the only beta-blocker able to limit the organ damage inflicted by hyperactivated neutrophils. These findings could have an impact on the treatment of diseases in which injury is linked to neutrophil hyperactivation, including sepsis and possibly even COVID-19.
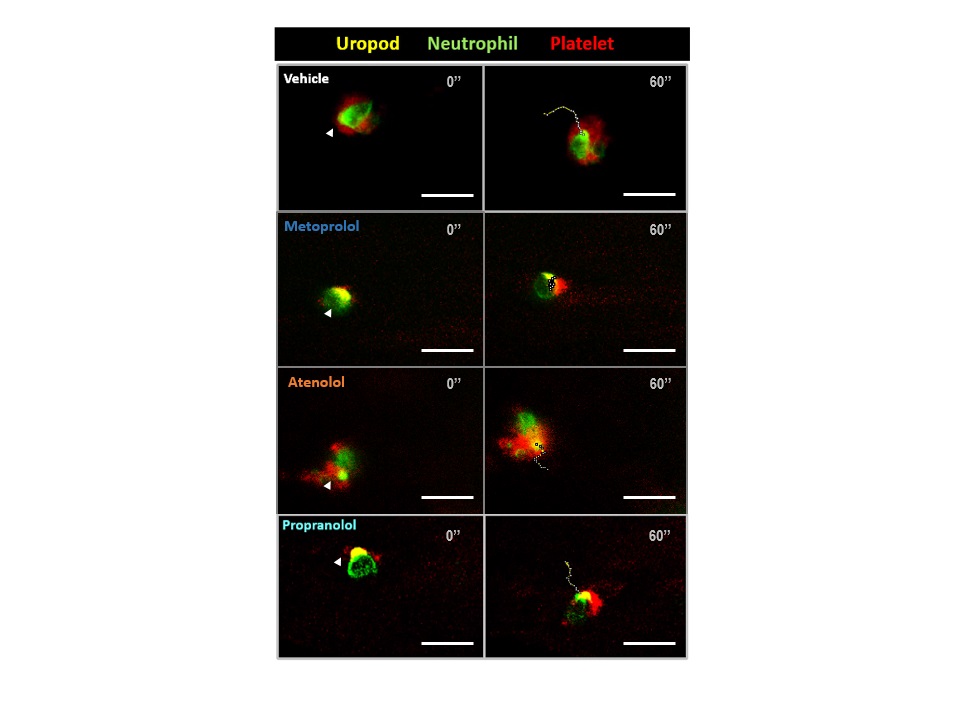
Building on the knowledge generated about metoprolol’s cardioprotective effect, the research team used 3D intravital microscopy to investigate the effect of the drug on neutrophils. “This advanced imaging technology allowed us to study changes in cell movement and shape induced by metoprolol and to exclude a direct effect on these cells of other intravenous beta-blockers like atenolol or propranolol,” explained Agustín Clemente.
Metoprolol, unlike other drugs in the same class, prevents inflammation after a myocardial infarction, thereby protecting heart function.
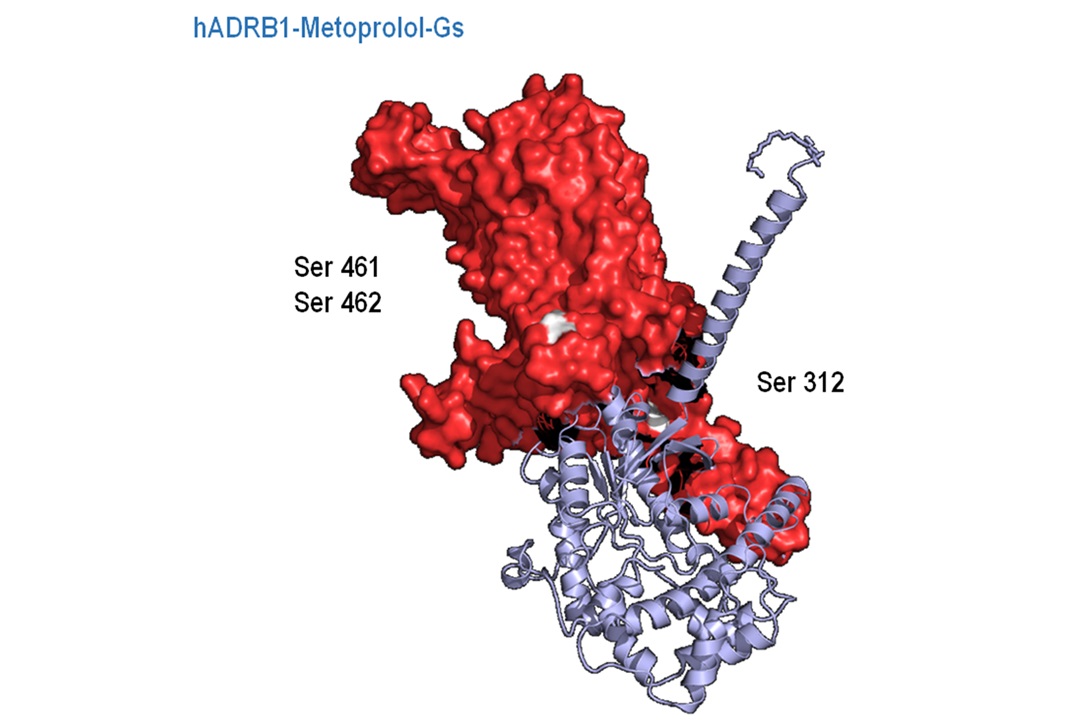
Working with the CNIC Bioinformatics Unit, the research team conducted computational studies to assess the impact of different beta-blockers on type 1 beta adrenergic receptors, the molecular target of these drugs. “Unexpectedly, we found that despite their related chemical structures each drug interacts with beta-1 adrenergic receptors in a different way. This translates into a distinct structural change in the receptor for each drug, which in the case of metoprolol induces a specific molecular cascade and a concrete and unique cellular effect,” commented co-principal investigator Dr Eduardo Oliver, a CNIC and CIBERCV pharmacologist and beneficiary of the Comunidad de Madrid talent program.
“Unlike other beta-blockers, metoprolol triggers a change in the intracellular structure of the beta-1 adrenergic receptor, opening it up so that it can interact with other nearby proteins that mediate the unique effect of metoprolol on neutrophil activity,” added Dr Oliver.
Until now, beta-blockers were thought to act exclusively by impeding interaction between epinephrine and beta-1 adrenergic receptors. But the new results demonstrate that the binding of metoprolol not only blocks the action of epinephrine, but also activates other intracellular pathways, a phenomenon called biased agonism.
The authors conclude that metoprolol should be the beta-blocker of choice in clinical practice. “If these results are confirmed in future clinical studies, this would herald a change in the clinical guidelines for this devastating disease, placing metoproplol, and not other beta-blockers, as the drug of choice for patients suffering a heart attack,” said Dr Ibañez.
The low cost of metoprolol, at less than 2 euros for an acute dose, brings additional value to this discovery.