Nature Communications: Identify a new mechanism involved in atherosclerosis
The study, published in Nature Communications, reveals these cells as a possible target for cardiovascular cell therapy
Researchers at the Centro Nacional de Investigaciones Cardiovasculares Carlos III (CNIC) have identified a new mechanism involved in the development of atherosclerosis. The study, published in Nature Communications indicates that nestin+ cells guide the inflammatory response in diseases affected by chronic inflammation, and therefore could be a new therapeutic target in these conditions. .
Atherosclerosis arises from fatty deposits in the blood vessel walls of arteries carrying oxygen-rich blood from the heart to the rest of the body. The disease has a high prevalence worldwide and is a major originating cause of cardiovascular events. Study leader Dr. Simón Méndez-Ferrer explained that atherosclerosis is a chronic inflammatory condition that begins when high levels of blood cholesterol activate the layer of endothelial cells lining the blood vessel. Study first author Dr. Raquel del Toro said: “This activation induces an infiltration of the vessel wall by inflammatory cells, forming the atherosclerotic plaque. When the plaque is weakened, sometimes due to calcification, it can rupture, releasing its contents to the bloodstream and starting a chain reaction that ends in the formation of one or more clots that block blood vessels”.
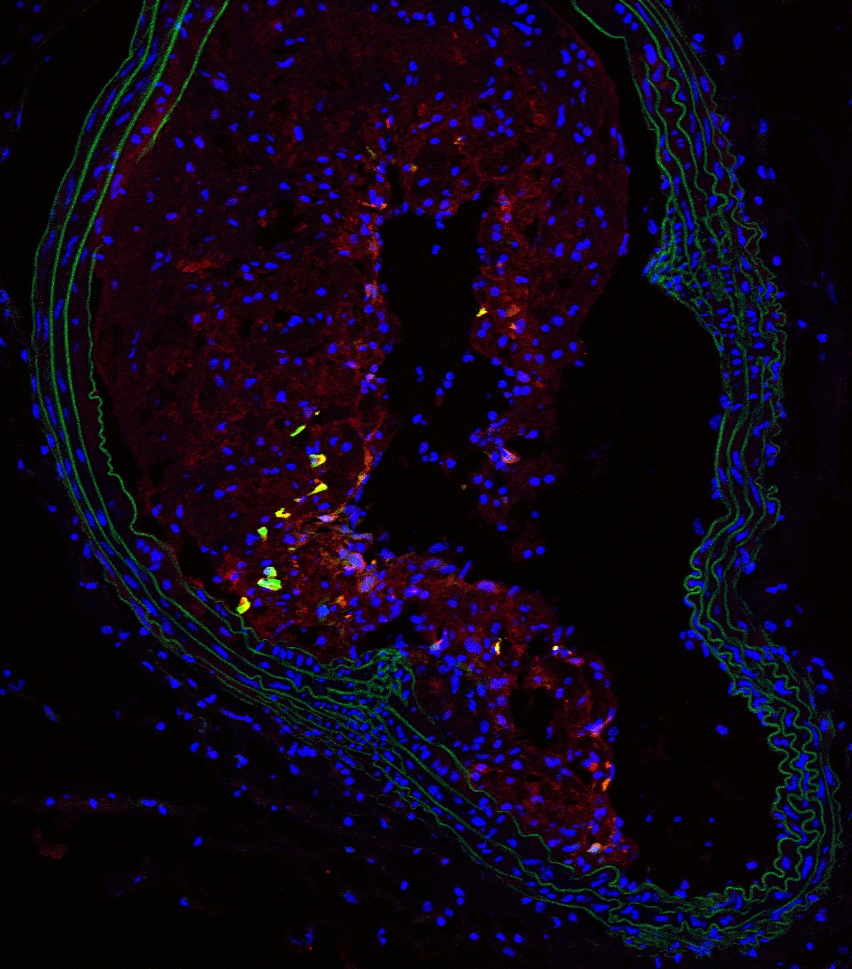
Cells expressing the protein nestin (nestin+) regulate inflammatory cell traffic toward the vessel wall during atherosclerosis
Most circulating inflammatory cells originate in the bone marrow, site to a host of cell types. Dr. Méndez-Ferrer said: “We know that nestin+ mesenchymal stem cells, together with endothelial cells, regulate the exit of inflammatory cells from the bone marrow into the blood in situations of acute inflammation, for example during bacterial infection.” The CNIC research team therefore decided to investigate if these cells also regulate inflammatory cell traffic during chronic inflammation, as produced in atherosclerosis.
The study used genetically modified mice in which nestin+ mesenchymal cells or endothelial cells specifically lack Mcp1, an important molecule in the migration of inflammatory cells to the circulation. Using these mice, the research team was able to show that nestin+ cells play an important role in the initiation of cardiovascular disease by regulating the migration of inflammatory cells from the bone marrow. The investigators also discovered nestin+ cells in the blood vessel wall that control inflammatory cell migration in this territory.
The results of the study suggest that nestin+ cells in the vessel wall contribute to the formation of the atherosclerotic plaque by promoting entry of inflammatory cells from the bloodstream. The key finding, in the researchers’ words, is that “elimation of Mcp1 in nestin+ cells, but not in endothelial cells, slows the progression of atherosclerosis and vascular calcification.”
The study reveals a new mechanism through which a subset of mesenchymal cells located in different organs guide inflammatory cells during chronic inflammation. These results highlight the potential of nestin+ cells as a therapeutic target in cardiovascular cell therapy.
"Nestin+ cells direct inflammatory cell migration in atherosclerosis”. doi:10.1038/ncomms12706