JACC: A drug costing less than €2 a day helps in the treatment of severely ill COVID-19 patients
The safety, availability, and robust biological effect of metoprolol could together be enough to consider its use in young patients admitted to the ICU with severe COVID-19
Metoprolol, a drug widely used to treat cardiovascular disease, is beneficial when administered to COVID-19patients. This is the finding of a study by investigators at the Centro Nacional de Investigaciones Cardiovasculares (CNIC), published today in the Journal of American College of Cardiology (JACC).
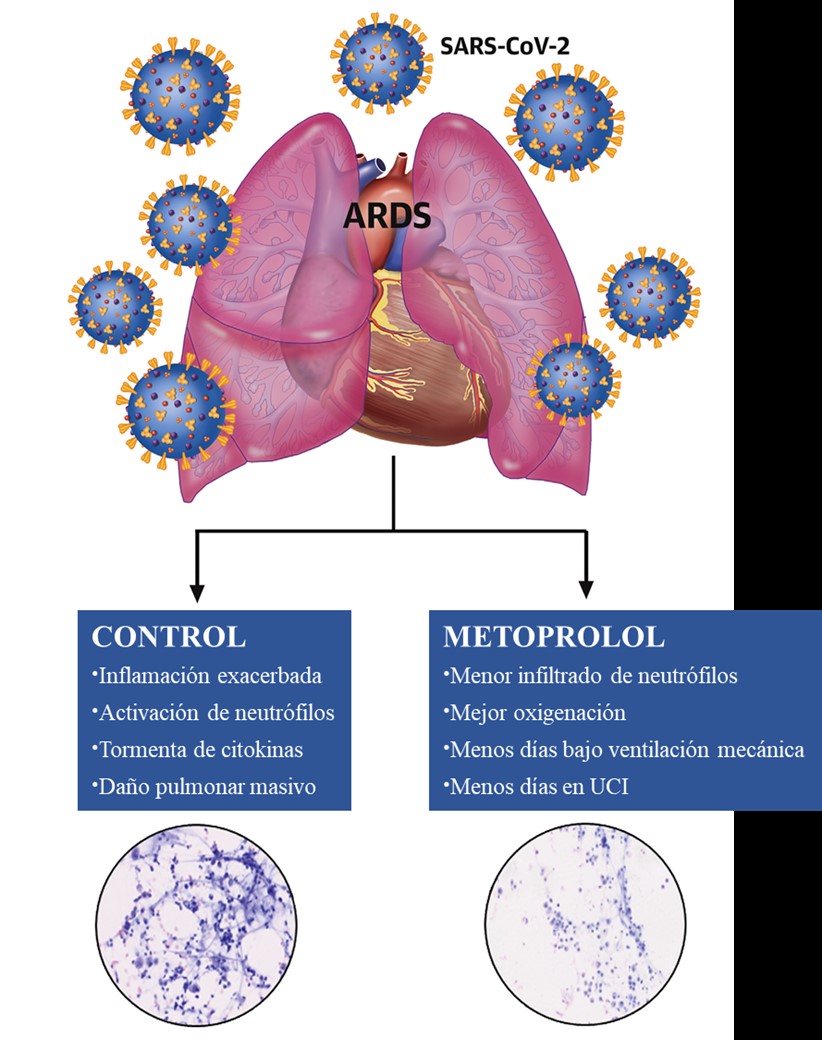
The most severe form of COVID-19 is severe respiratory failure, which requires intubation and is associated with a high mortality rate. Pulmonary infection with the SARS-CoV2 virus can progress to acute respiratory distress syndrome (ARDS), in which inflammation and neutrophil hyperactivation play a central role. There is currently a lack of therapies for ARDS associated with COVID-19.
The study was led by Dr. Borja Ibáñez, group leader of the Translational Laboratory for Cardiovascular Imaging and Therapy at the CNIC, cardiologist at the Hospital Universitario Fundación Jiménez Díaz (FJD) in Madird, and member of the CIBERCV cardiovascular research network. The research team recently discovered that metoprolol, a well-established beta-blocker, has a highly selective effect on hyperactivated neutrophils during situations of acute stress such as a myocardial infarction. Given the central role played by neutrophils in ARDS, the team postulated that metoprolol might be an effective treatment for patients with severe COVID-19.
MADRID-COVID is a randomized clinical trial conducted in close collaboration between the CNIC and cardiology, ICU, pulmonology, and biobank services at FJD Hospital. This pilot trial examined the effect of intravenous metoprolol administration on lung inflammation and respiratory function in severe COVID-19 patients intubated after developing ARDS.
Describing the study, Dr. Ibáñez explained that that the team “randomized 20 intubated COVID-19 patients to receive intravenous metoprolol (15 mg per day over 3 days) or to a control group that did not receive metoprolol. We analyzed the inflammatory infiltrate in bronchoalveolar fluid before and after treatment and also monitored clinical progression parameters such as oxygenation and days on mechanical ventilation.”
Co-author Eduardo Oliver, a Ramón y Cajal fellow at the CNIC, affirmed that the intravenous metroprolol treatment “significantly reduced neutrophil infiltration of the lungs and improved oxygenation.” Intensive care specialist Arnoldo Santos, who coordinated the ICU part of the study, indicated that the team saw “a clear trend among the metoprolol-treated patients to require fewer days on mechanical ventilation and therefore a shorter stay in the ICU.”
Dr. Ibáñez added that “while we need to be cautious with these results of a pilot trial, we have observed that metoprolol treatment in this clinical setting is safe, is associated with a very significant reduction in lung infiltration, and appears to lead to very rapid improvements in patient oxygenation.”
The researchers therefore propose intravenous metoprolol as a “promising intervention that could improve the prognosis of severely ill COVID-19 patients.” They also emphasize that metoprolol is a safe and cheap drug (daily treatment cost below €2) that is readily available.
Joint first author Agustín Clemente-Moragón added that “the effect of metoprolol on the hyperactivation of inflammatory cells implicated in ARDS is exclusive to this beta-blocker.” In a previous experimental study, the same group recently demonstrated that other apparently similar beta-blockers have no effect on exacerbated lung inflammation.
The study is a prime example of collaboration between basic and clinical researchers. Indeed, the first 4 authors made equal contributions to the study and are 2 basic researchers (Clemente-Moragón and Oliver), a cardiologist (Martínez-Milla), and an intensive care specialist (Santos).
Dr. Valentín Fuster, CNIC General Director and co-investigator on this study, indicated that “although this was a pilot study and will need to be confirmed in a larger trial, the safety, availability, and robust biological effect of metoprolol could together be enough to consider its use in young patients admitted to the ICU with severe COVID-19”.
The research team led by Dr. Ibáñez was recently awarded funding from the Instituto de Salud Carlos III (ISCIII) for a clinical trial to definitively demonstrate the clinical benefits of metoprolol in 350 ADRS patients admitted to 14 ICUs across Spain. The MAIDEN clinical trial will be coordinated by the cardiovascular CIBER research network and will include the participation of cardiovascular and respiratory specialists.
The study was partly funded by the European Comission (EERC-CoG grant Nº 819775) and the Ministerio de Ciencia e Innovación (MCN; ‘RETOS 2019’ grant Nº PID2019-107332RB-I00). The study was also supported by the Programa de Atracción de Talento de la Comunidad de Madrid.