Nature Immunology: Clustering for health
When functioning appropriately, the immune system protects us from multiple threats such as pathogens, disease-causing microbes, and tumors. However, when the immune system is inappropriately activated, it attacks our own body, causing autoimmune syndromes. In the classical view, autoimmunity is considered as a result of an hyperactive immune system. However, it has recently become clear that autoimmunity can also occur as a result of a weakened immune response. This new view is crucial for the strategy of therapeutic intervention: immune suppression versus immune stimulation. Within the framework of this new concept, Dr. Susana Minguet and colleagues searched for models in which a weakened immune response was associated to autoimmunity, thus identifying the membrane protein Caveolin-1 as a key regulator of this paradoxical scenario.
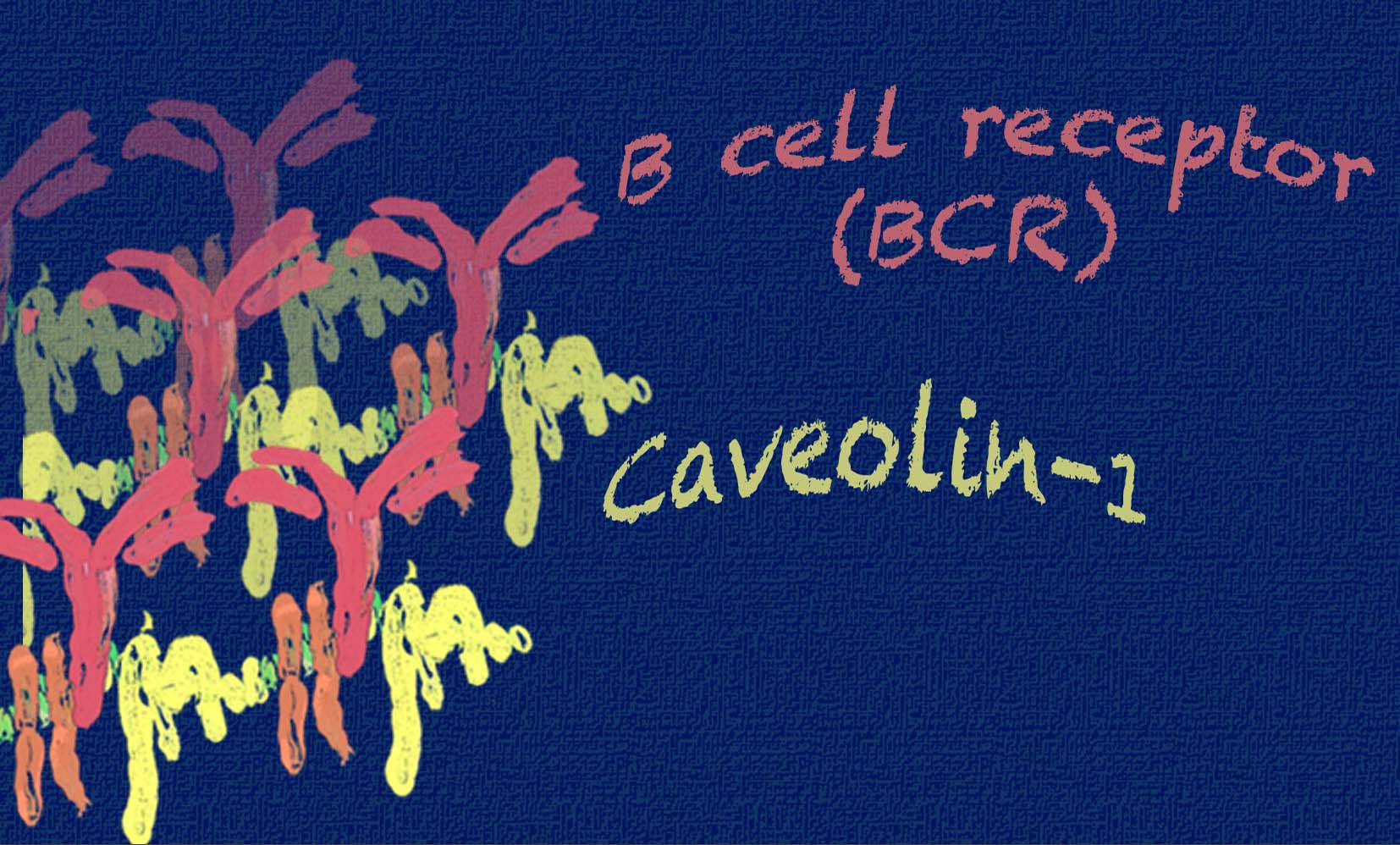
The team of scientists identified the protein Caveolin-1 as a crucial regulator of the organization of the plasma membrane of B cells. B cells recognize pathogens using the B cell antigen receptor (BCR), a receptor located at the cell surface, linking the extracellular space- where most pathogens are recognized- with the intracellular space. Thus, the BCR works as an “antenna” that allows B cells to sense infections, become activated, and eliminate the threat. Recent work from Prof. Reth, also coauthor in this study, and Dr. Batista (Boston, USA) has demonstrated that these antennas are not randomly distributed on the surface of B cells, but highly organized in protein islands which coalesce when the BCR recognizes a threat. Now, Dr. Susana Minguet and co-workers have demonstrated that the protein Caveolin-1 regulates this organization, which is crucial for an efficient transmission of the activating signals to the B cells, in order to mount an immune response against the pathogen. Thus, in the absence of Caveolin-1, B cells do not sense the threat properly, thus hampering a proper immune response.
The authors went one-step forward, demonstrating that the deficient activation of the BCRs also led to autoimmune disease in animal models. Newly generated B cells have to be educated to be blind to self-molecules, while still being able to recognize and become activated by pathogenic molecules. This mechanism is known as B cell tolerance and relies in the efficient transmission of signals by the BCR. In this study, Dr. Susana Minguet and colleagues demonstrated that newly generated B cells lacking the protein Caveolin-1 do not properly organize their BCR on the cell surface and fail to transmit the signals needed to establish tolerance. Thus, B cells lacking the protein Caveolin-1 are not blind to self-molecules, recognizing them as a threat instead, thus becoming activated and triggering an active immune response against self, i.e. autoimmunity.
These results, published by Nature Immunology, have the potential to improve our current understanding and treatment of autoimmune disorders, since the scientific community at present lacks proper models recapitulating the autoimmunity that is associated with immunodeficiency as observed in patients.
Dr. Susana Minguet started the project in 2008 in the laboratory of Dr. Miguel Angel Del Pozo at the Centro Nacional de Investigaciones Cardiovasculares Carlos III supported first by a MINECO grant and later by the Ramón y Cajal program from the Spanish Ministry of Economy and Competitiveness. In July 2011, Dr. Minguet moved to the University of Freiburg as an independent group leader, pursuing this project under the umbrella of the Cluster of Excellence BIOSS Centre for Biological Signalling Studies and the Center for Center for Chronic Immunodeficiency (CCI), and the support of the Collaborative Research Center 1160 "Immune-mediated pathology as consequence of impaired immune reactions (IMPATH)" at the University of Freiburg, and in close collaboration with the groups of Prof. Miguel A. Del Pozo and Prof. Michael Reth.
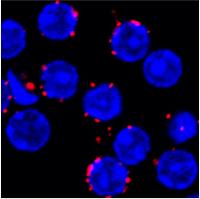
En linfocitos B (en azul en la imagen de microscopía confocal), la proximidad entre receptores (BCRs) se analiza mediante una técnica llamada PLA (del inglés, “Proximity Ligation Assay”) que genera señales fluorescentes en rojo cuando los receptores están una distancia menor de 20 nanómetros. Autor: Kathrin Kläsener