Circulation Research: CNIC scientists describe one of the mechanisms underlying cardiac hypertrophy
The new findings, published in Circulation Research, could spur the development of new tools for the treatment of cardiac hypertrophy
Scientists at the Centro Nacional de Investigaciones Cardiovasculares (CNIC) have identified a new mechanism involved in the regulation of cardiac hypertrophy. The mechanisms underlying this disease remain largely unknown, and this situation is impeding the development of effective treatments.
The findings, published in Circulation Research, may spur the development of new tools for the treatment of cardiac hypertrophy, especially in Cushing syndrome patients.
The results, explained principal investigator Dr. Enrique Lara Pezzii, who leads the Molecular Regulation of Heart Failure group at the CNIC, reveal that the “protein SRSF4 binds to and stabilizes the non-coding RNA GAS5, enabling it to block the action of the glucocorticoid receptor and thus prevent cardiac hypertrophy”.
The contractile function of the heart depends on the action of cardiomyocytes. Although in the adult heart these cells have almost no ability to divide, Dr. Lara Pezzi explained that they have a remarkable ability to adapt to the changing demands placed on the heart.
One example of this capacity is the response to aortic stenosis. “During stenosis, the aortic valve cannot open fully, and the narrowing of the opening obliges the left ventricle to pump blood with greater force. This requires the heart muscle to contract more strongly,” continued Dr. Lara Pezzi.
Although cardiomyocytes cannot divide, Dr. Lara Pezzi explained that “to increase contractile capacity, these cells become enlarged, a process known as hypertrophy.”
However, while this response is effective at first, the thickening of the left ventricular wall (cardiac hypertrophy) triggers structural changes in the heart that cause a progressive loss of contractile capacity.
Continuing lack of knowledge about the mechanisms underlying cardiac hypertrophy is impeding the development of effective treatments
Dr. Lara-Pezzi’s group, in partnership with researchers at Hospital Puerta de Hierro in Majadahonda, the Spanish Cardiovascular Research Network (CIBERCV), and the University of Frankfurt, has analyzed possible mechanisms underlying the development of this disease.
The researchers found that mice lacking the RNA-binding protein SRSF4 develop cardiac hypertrophy and have an impaired ability to relax the heart muscle, a condition known as diastolic dysfunction.
Further analysis showed that the absence of SRSF4 severely reduces the expression of the non-coding RNA GAS5. “SRSF4 binds directly to GAS5, preventing GAS5 degradation in the cell. GAS5 is an inhibitor of the glucocorticoid receptor, whose activation contributes to the development of cardiac hypertrophy,” explained Dr. Lara Pezzi.
First author Dr. Javier Larrasa clarified that “the absence of SRSF4 triggers the degradation of GAS5, and this results in activation of the glucocorticoid receptor. In contrast, overexpression of GAS5 with a viral vector inhibits the glucocorticoid receptor and reduces cardiac hypertrophy.”
.
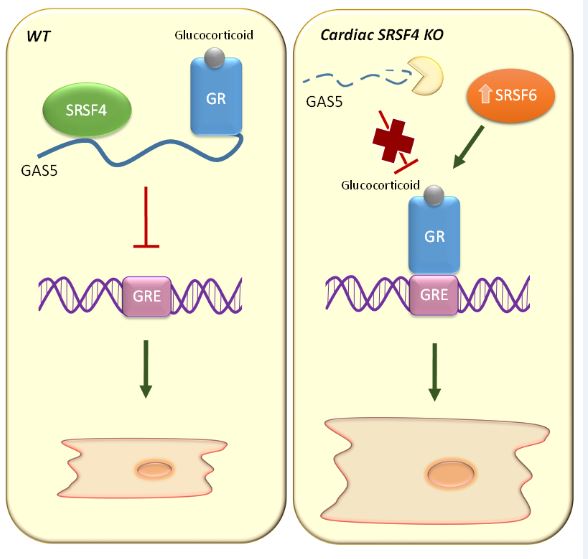
The research team concludes that the identification of the SRSF4–GAS5–glucocorticoid-receptor cell signaling pathway is a major advance in the characterization of the molecular mechanisms underlying cardiac hypertrophy and could serve as the basis for new treatments.
Furthermore, the findings could be “particularly applicable to patients with Cushing syndrome, who develop cardiac hypertrophy in part through glucocorticoid receptor activation.”
An analysis of samples from Cushing syndrome patients revealed a degree of dysregulation of SRSF4 and GAS5 expression; however, the possible role of this pathway in this disease will require further research.
The study received funding from the European Union (CardioNeT-ITN-289600 and CardioNext-ITN-608027), the Ministerio de Economía y Competitividad, and the Community of Madrid (2010-BMD-2321 “Fibroteam”). Funding was also provided by the Plan Estatal de I+D+I 2013-2016 and the European Regional Development Fund “A way to build Europe” program.











