Jose Luis Cabrera Alarcón, PhD

Over seven years as a postdoctoral researcher in the GENOXPHOS group, we have studied the OxPhos system from an evolutionary perspective integrated with its three-dimensional structure. This approach enables us to bridge molecular evolution with spatial organization and functional constraints. By adopting this methodology, we not only leverage phylogenetic insights from other groups to make clinical predictions but also assess the relative significance of specific positions or clusters of positions within OxPhos with greater precision.

Marta Pérez-Hernández Durán, PhD

After completing my PhD in Cardiac Electrophysiology at the Complutense University of Madrid, I carried out a first postdoctoral stage at New York University (USA), where I studied the function of cardiomyocytes under physiological conditions and in the context of arrhythmogenic right ventricular cardiomyopathy (ARVC). I later joined the GENOXPHOS group to investigate the role of the kinase Fgr in the heart.
Our studies have shown that Fgr becomes activated under cellular stress and, within mitochondria, phosphorylates complex II of the electron transport chain, triggering a signaling cascade that promotes inflammation. My current project aims to determine whether Fgr inhibition exerts a cardioprotective effect by reducing cardiac inflammation.
To address this question, we use several experimental models (including Fgr-deficient mice) in the context of aging, exercise, myocardial infarction, atherosclerosis, and ARVC, among others. Within this framework, we maintain several active collaborations:
- With New York University, through a La Caixa Health Research Project, we are evaluating whether Fgr inhibition reduces inflammation in a murine model of ARVC.
- With the Hospital Universitario de Getafe, we study the role of Fgr in patients with myocardial infarction with non-obstructive coronary arteries (MINOCA), within the scope of a FIS-funded project.
- In addition, we collaborate with the company Nostrum in the development and characterization of novel, specific Fgr inhibitors, aiming to explore their therapeutic potential in inflammatory cardiac diseases.
Finally, I lead a “Generación de Conocimiento” (Knowledge Generation) project focused on characterizing the role of mitochondria in ARVC, with the goal of identifying new molecular mechanisms involved in this pathology.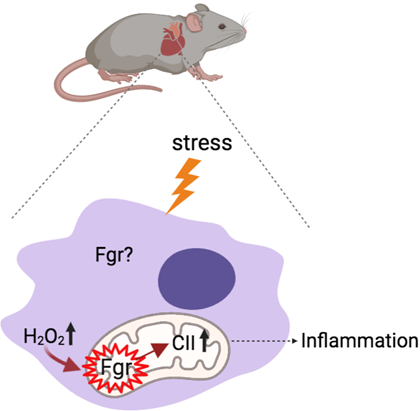
María Concepción Jiménez Gómez, PhD

I hold a PhD in Molecular Biology from the Autonomous University of Madrid (UAM) and, since 2010, I have worked at the Spanish National Cardiovascular Research Center (CNIC) within the Genoxphos group, led by Dr. Enríquez. My work at the CNIC has focused primarily on the comprehensive management of research projects and the promotion of translational development initiatives.
Within the Genoxphos group, I have been responsible for the planning, coordination, and supervision of scientific projects, including managing timelines, resources, budgets, and relationships with national and international collaborators. I also coordinate strategic alliances with the pharmaceutical industry, including collaborations with AstraZeneca and Minovia Therapeutics, playing a key role in scientific and technical communication and in aligning objectives among the teams involved.
My commitment to efficient management led me to complete two master's degrees in management and administration, which have enabled me to optimize the group's internal processes and strengthen strategic decision-making. I am currently pursuing a degree in Psychology, with the aim of integrating knowledge of comparative and behavioral psychology into the design and execution of scientific projects.
Alongside my management work, I participate in research projects focused on the role of the mitochondrial metalloproteinase OMA1 as an integrator of cellular stress, in collaboration with Dr. Yolanda Martí. Our studies have revealed that the deletion of OMA1—traditionally considered protective in certain pathological contexts—can promote the development of liver pathologies in Oma1KO mouse models, including increased mortality, predisposition to primary tumors, and progression from early liver damage to chronic inflammation and fibrosis. These results suggest an essential role for OMA1 in protecting against chronic liver disease and tumorigenesis by regulating hepatic immunogenicity.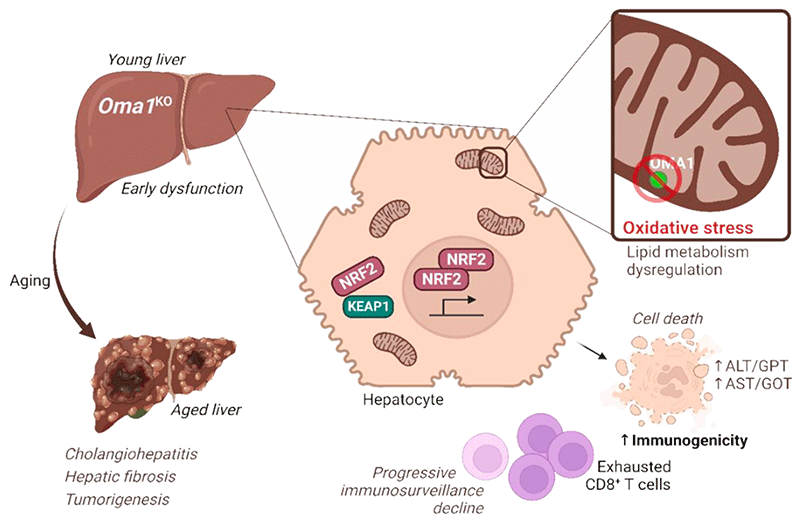
Raquel Justo Méndez
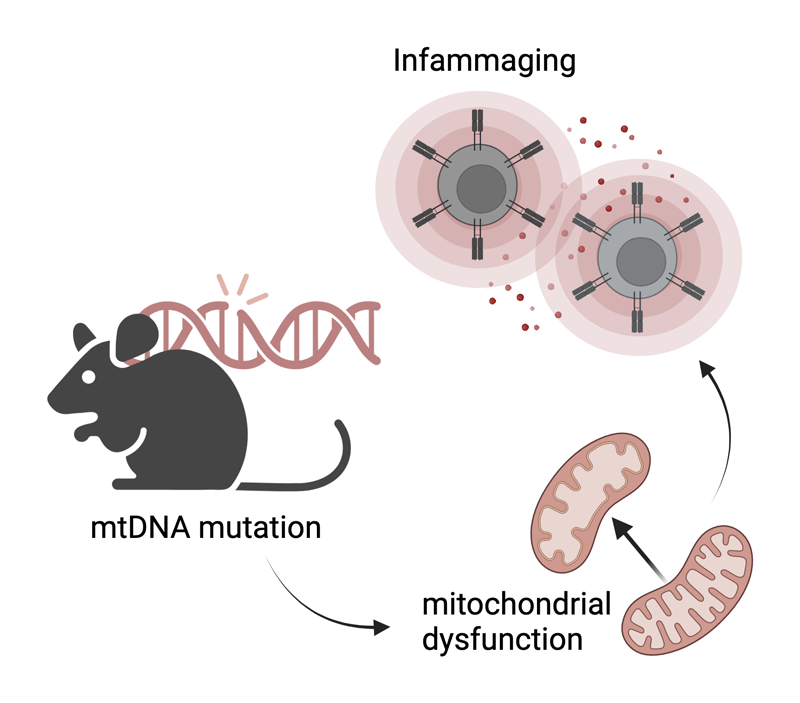
I am biologist (Universidad Autónoma de Madrid) currently performing my phD in Molecular Biomedicine under the supervision of José Antonio Enríquez and Ana Victoria Lechuga-Vieco in which we are evaluating the impact of mito-nuclear crosstalk during hematopoiesis under conditions of normal vs. high mitochondrial variability. Our study involves immune, metabolic, and mitochondrial profiling, focusing on aspects such as ROS management, organization of respiratory complexes, and fuel preference.
By using a mouse model deficient in the proofreading capacity of the mitochondrial DNA polymerase we shed light on how immune cells contribute to inflammaging, frailty and age-related phenotypes, emphasizing the significance of mitochondrial quality control and translation mechanisms.
Carmen Morales Vidal
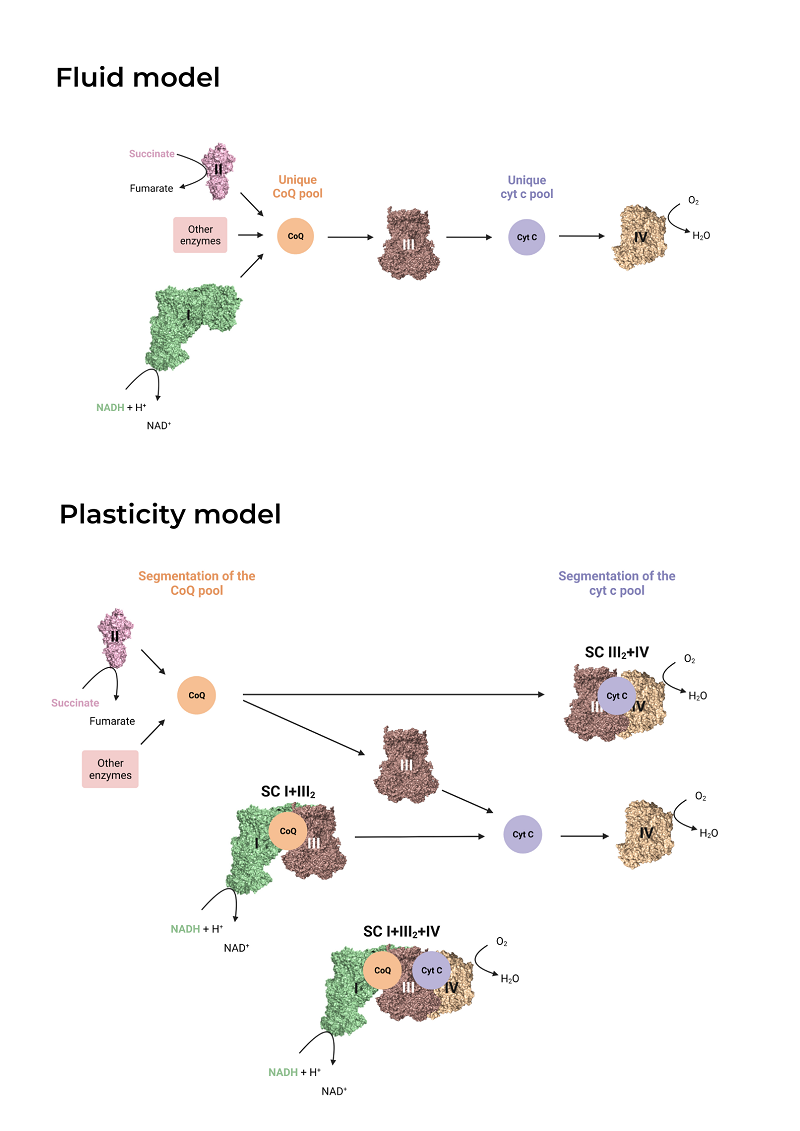
 After graduating in Biochemistry and Biomedical Sciences from the University of Valencia in 2020, I joined the Genoxphos group as a master’s student and later as a predoctoral researcher. I am currently finishing writing my thesis. My project focuses on understanding the reason behind the superassembly of mitochondrial complexes I (CI) and III (CIII), an interaction that is highly conserved in nature, from yeast to mammals, and gives rise to supercomplexes such as I+III₂ and I+III₂+IV. Several functions have been proposed for this association, including substrate channeling between complexes, reduction of reactive oxygen species production, and stabilization of the constituent complexes. To study the role of the I+III2 interaction in mitochondrial bioenergetics and morphology in more detail, I have generated genetically modified cell lines in which the interaction between CI and CIII is disrupted. This has allowed me to observe that, in the absence of I+III2 superassembly, mitochondria exhibit reduced basal and maximal respiration, produce more reactive oxygen species, and are more sensitive to substrate changes, among other effects. These models fill a gap in the field, as prior to this thesis, no living system with a loss of CI-CIII association was available—an essential tool to clarify its controversial role in living organisms.
After graduating in Biochemistry and Biomedical Sciences from the University of Valencia in 2020, I joined the Genoxphos group as a master’s student and later as a predoctoral researcher. I am currently finishing writing my thesis. My project focuses on understanding the reason behind the superassembly of mitochondrial complexes I (CI) and III (CIII), an interaction that is highly conserved in nature, from yeast to mammals, and gives rise to supercomplexes such as I+III₂ and I+III₂+IV. Several functions have been proposed for this association, including substrate channeling between complexes, reduction of reactive oxygen species production, and stabilization of the constituent complexes. To study the role of the I+III2 interaction in mitochondrial bioenergetics and morphology in more detail, I have generated genetically modified cell lines in which the interaction between CI and CIII is disrupted. This has allowed me to observe that, in the absence of I+III2 superassembly, mitochondria exhibit reduced basal and maximal respiration, produce more reactive oxygen species, and are more sensitive to substrate changes, among other effects. These models fill a gap in the field, as prior to this thesis, no living system with a loss of CI-CIII association was available—an essential tool to clarify its controversial role in living organisms.
Raquel Martínez de Mena, PhD
 After obtaining my PhD in Science from the Autonomous University of Madrid and before joining Dr. José Antonio Enríquez's team, I conducted research on brown adipose tissue and thyroid hormones at the Alberto Sols Biomedical Research Institute (IIB) in Madrid.
After obtaining my PhD in Science from the Autonomous University of Madrid and before joining Dr. José Antonio Enríquez's team, I conducted research on brown adipose tissue and thyroid hormones at the Alberto Sols Biomedical Research Institute (IIB) in Madrid.
For the past eight years at the CNIC, I have contributed my expertise in biochemical and molecular biology techniques, focusing on the functional genetics of the oxidative phosphorylation system.
I actively participate, both scientifically and technically, in the group's projects related to mitochondrial metabolism and physiology using cell and mouse models.
María del Mar Muñoz Hernández

Me uní al grupo GENOXPHOS en 2014, tras finalizar mi formación como técnico de laboratorio en Anatomía Patológica y Citología.
Dentro del grupo, participo activamente en diversas líneas de investigación, con especial dedicación al manejo de modelos animales experimentales. Mi labor abarca la cría y caracterización genética, además de contar con formación específica en técnicas quirúrgicas —principalmente cardíacas— y en la realización de análisis metabólicos y de comportamiento vinculados al estudio del envejecimiento.

Eva Raquel Martínez

As a laboratory technician, I am responsible for conducting experiments, analyzing samples, and performing tests in a laboratory. My role often involves using various scientific instruments and ensuring accurate recording of data for research or diagnostic purposes.
These techniques range from DNA genotyping to the treatment of tissue samples with fine diagnostics.
Marina Rosa Moreno
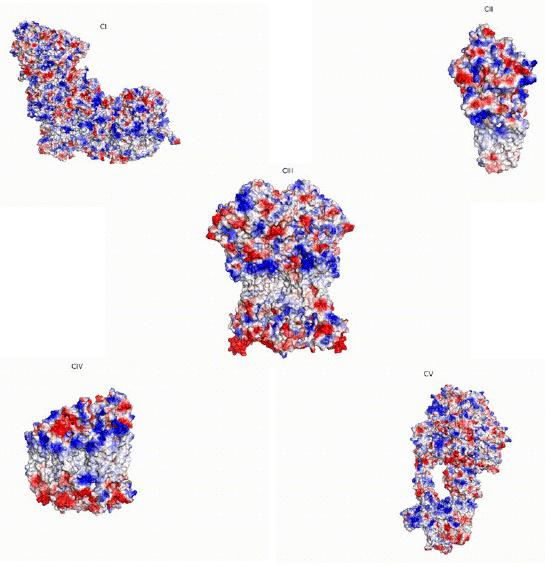
 My project focuses on the structural analysis of the mitochondrial respiratory chain complexes and supercomplexes. I combine in silico computational modeling and the use of cryoelectron microscopy as a structural determination technique. Also, we are able to integrate the models obtained through these approaches with genetic information from public databases, which allows us to develop more accurate pathogenicity predictions and to carry out comparative studies that help us better understand the evolutionary processes involved in diseases as well as in speciation events or allele selection.
My project focuses on the structural analysis of the mitochondrial respiratory chain complexes and supercomplexes. I combine in silico computational modeling and the use of cryoelectron microscopy as a structural determination technique. Also, we are able to integrate the models obtained through these approaches with genetic information from public databases, which allows us to develop more accurate pathogenicity predictions and to carry out comparative studies that help us better understand the evolutionary processes involved in diseases as well as in speciation events or allele selection.

Paula Fernández-Montes Díaz

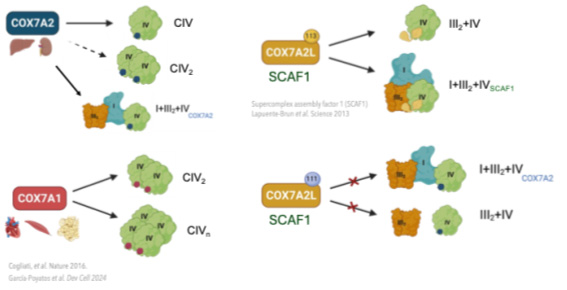
After graduating in Biology at the Universidad Autónoma de Madrid, I joined the GENOXPHOS group as a master student and finally as a predoctoral researcher. In my third year, I study the role of alternative isoforms of complex IV, specifically the Cox7a family. Complex IV (cytochrome oxidase c) is the last complex in the electron transport chain and undergoes a high level of regulation. The COX7A family is formed by different isoforms: COX7A1, COX7A2 and COX7A2L or SCAF1 and they are interchangeable. The alternative Cox7a subunits confer to complex IV different supramolecular organization, appearing in monomeric, dimer, multimer and super-assembled with other complexes forming the Q-respirasome and the N-respirasome (Cogliati and Calvo, et al. Nature 2016).
Using murine and cellular models, we have observed that the supramolecular structures of complex IV have different metabolic and physiological functions, ranging from organism viability, metabolic maturation necessary for proper tissue physiology, to plasticity and adaptation to different metabolic requirements.
Michaela Veliova, PhD

I completed my master’s degree in Biotechnology in Vienna, Austria and went on to earn my Ph.D. in Molecular and Medical Pharmacology at the University of California, Los Angeles (UCLA). As a postdoctoral researcher in the GENOXPHOS lab, my work investigates the intracellular diversity of cardiomyocyte mitochondria, focusing on the functional differences between interfibrillar mitochondria (IFM) and subsarcolemmal mitochondria (SSM), two mitotypes with distinct roles in cardiomyocyte physiology. My current research explores how the subcellular environment, particularly cytoplasmic ribosomes, contributes to mitotype differentiation, aiming to understand how differential protein expression is regulated and how the cellular microenvironment shapes mitochondrial proteomes and function. Through this work, I seek to uncover the principles driving mitochondrial specialization and diversity within cardiomyocytes, and to understand how these functional specializations change during disease or aging.
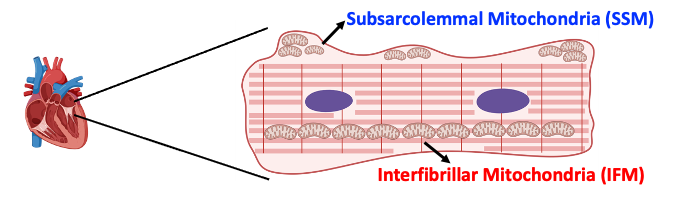
Rebeca Acín

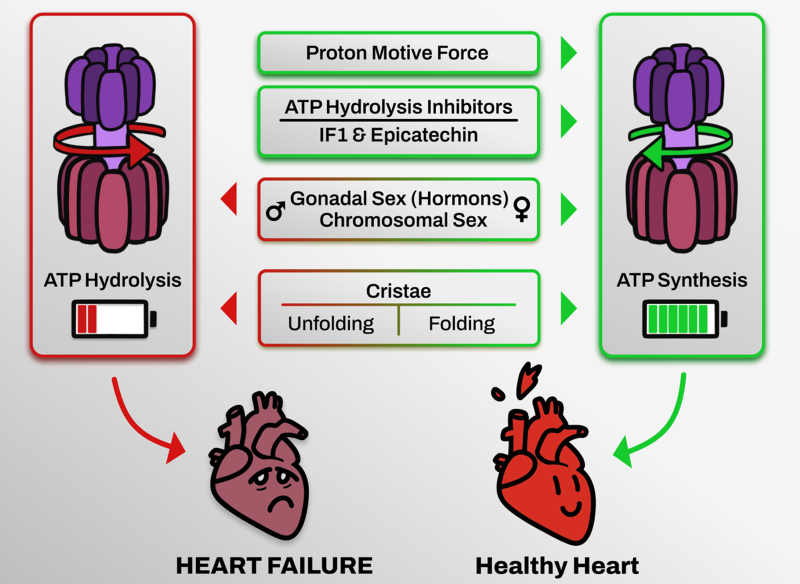
My line of research focuses on studying the role of mitochondria in heart failure. In particular, my project examines how ATP depletion—caused by the reverse operation of Complex V or the mitochondrial ATPase—contributes to the adverse phenotype associated with cardiac dysfunction. Recent work from our group has demonstrated that inhibiting this reverse activity of Complex V has a beneficial effect, improving mitochondrial performance under stress or mitochondrial pathology. Extrapolating these findings to heart failure models will allow us to identify new therapeutic targets and strategies to slow the progressive deterioration of the heart in cardiovascular diseases.
From a more molecular perspective, my project aims to investigate the different assembly forms of Complex V in order to elucidate the role each one plays in ATP synthesis or hydrolysis. To this end, we have access to animal and cellular models carrying assembly-deficient mutations in Complex V.
Sara Natalia Jaroszewicz

I graduated in Biochemistry from the Autonomous University of Madrid in 2022 and joined the group in the summer of that same year as a CICERONE internship student and later as a Master's student. My doctoral thesis consists of deciphering the in situ organization of the electron transport chain using super-resolution techniques.
Mitochondria are essential organelles in eukaryotic cellular metabolism. Oxidative phosphorylation occurs in them and this process is mediated by respiratory complexes (RCs), which can assemble into macromolecular structures known as supercomplexes (SCs). Although the existence of SCs has been demonstrated, there are still doubts about how these complexes are actually organized in the inner mitochondrial membrane and how their proportion could influence the development of heart diseases. To answer these questions, my project focuses on the optimization of STED, Expansion Microscopy (iU-ExM) y proximity ligation assay (PLA) super-resolution methods in order to visualize SCs in situ, determine their physiological position in subdomains of the mitochondrial cristae and compare them quantitatively in a murine model of heart failure.

Carolina Amalia Simón Guerrero
After three years of experience as a Bioinformatician developing and maintaining scientific software and data analysis tools (primarily focused on Structural Biology and Cryo-Electron Microscopy), I joined the GENOXPHOS lab to contribute to the development of an integrated bioinformatics platform for comprehensive analysis of the OxPhos system’s pathophysiology.